 Back
BackChemical Kinetics: Reaction Rates and Rate Laws
Study Guide - Smart Notes

Chemical Kinetics
Introduction to Chemical Kinetics
Chemical kinetics is the area of chemistry concerned with the speed, or rate, at which chemical reactions occur. While thermodynamics tells us whether a reaction is favored, kinetics explains how quickly it proceeds. Understanding reaction rates is essential for predicting product formation and optimizing chemical processes.
Reaction Rates
The reaction rate measures how fast reactants are converted to products. It is defined as the change in concentration of reactants or products per unit time, typically expressed in units of M/s (mol/(L·s)). The rate of appearance of products is directly related to the rate of disappearance of reactants.
General formula:
Example: For the reaction A → B:
Stoichiometry and Reaction Rates
For reactions with non-1:1 stoichiometry, the rate must account for the coefficients in the balanced equation:
General formula:
Types of Reaction Rates
There are two main types of reaction rates:
Instantaneous Rate: The rate at a specific moment in time.
Average Rate: The rate over a period of time or the entire experiment.
Rate Laws
Definition and Formulation
The rate law (or rate equation) expresses the reaction rate as a function of reactant concentrations. It is determined experimentally and is useful for predicting reaction rates under various conditions.
General form:
k: Rate constant (depends on temperature)
m, n: Reaction orders (must be determined experimentally)
Determining the Rate Law: Method of Initial Rates
The rate law is determined by measuring initial rates at different reactant concentrations. The method of initial rates assumes known initial concentrations, zero initial product, and negligible reverse or competing reactions.
Vary the concentration of one reactant while keeping others constant.
Take the ratio of rates from two experiments to solve for reaction order.
Once orders are known, use experimental data to solve for the rate constant, k.
Example: Rate Law Determination
For the reaction of sodium hydroxide with methyl acetate:
Rate law found: First order in each reactant, overall second order.
Rate law:
Integrated Rate Laws
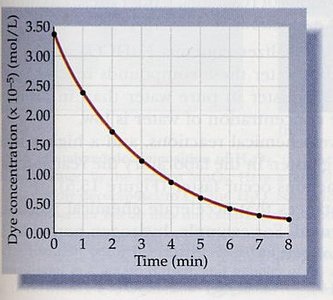
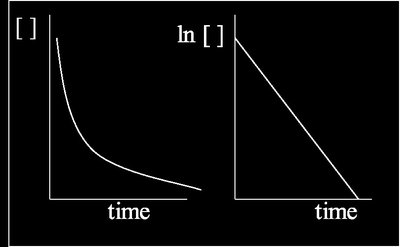
First Order Reactions
First order reactions have rates directly proportional to the concentration of one reactant. Radioactive decay is a classic example.
Rate law:
Integrated rate law:
Linear form:
Half-life:
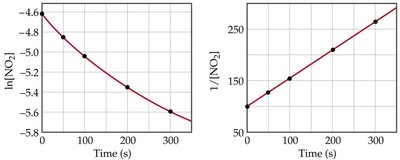
Second Order Reactions
Second order reactions depend on the square of the concentration of one reactant or the product of two reactants.
Rate law:
Integrated rate law:
Half-life:
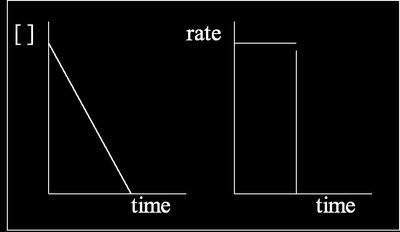
Zero Order Reactions
Zero order reactions proceed at a constant rate, independent of reactant concentration.
Rate law:
Integrated rate law:
Half-life:
Summary Table: Rate Laws and Integrated Rate Laws
The following table summarizes the key features of zero, first, and second order reactions:
Order | Rate Law | Integrated Rate Law | Plot (Straight Line) | Slope-Rate Constant Relationship | Half Life |
|---|---|---|---|---|---|
Zero | vs. | Slope = | |||
First | vs. | Slope = | |||
Second | vs. | Slope = |
Additional info:
Some examples and applications, such as radioactive decay and hydrogen peroxide decomposition, illustrate the use of integrated rate laws. The phase diagram of carbon (diamond, graphite, liquid, vapor) is referenced but is not directly relevant to chemical kinetics; it is more pertinent to thermodynamics and phase transitions.
