 Back
BackChemical Kinetics: Reaction Rates and Rate Laws
Study Guide - Smart Notes

Chemical Kinetics
Introduction to Reaction Rates
Chemical kinetics is the study of the speed at which chemical reactions occur and the factors that influence these rates. Understanding reaction rates is essential for controlling chemical processes in industrial, environmental, and biological contexts.
Reaction Rate: The change in concentration of a reactant or product per unit time.
Example: The conversion of diamond (C, s) to graphite (C, s) is extremely slow, while the decomposition of sodium azide (used in airbags) is very fast.



Everyday Examples: Fruit ripening and rotting, and the rapid color change in chemical reactions.


Factors Affecting Reaction Rates
The rate of a chemical reaction can be influenced by several factors:
Concentration: Higher concentrations of reactants generally increase reaction rates due to more frequent collisions.
Temperature: Raising the temperature increases the kinetic energy of molecules, leading to more effective collisions.
Catalysts: Catalysts lower the activation energy, increasing the rate without being consumed.
Surface Area: For reactions involving solids, increasing surface area increases the rate.
Measuring Reaction Rates
Reaction rates are typically measured by monitoring the change in concentration of a reactant or product over time. This can be visualized using concentration vs. time graphs.
Average Rate: Calculated over a time interval as the change in concentration divided by the change in time.
Instantaneous Rate: The rate at a specific moment, given by the slope of the tangent to the concentration vs. time curve.
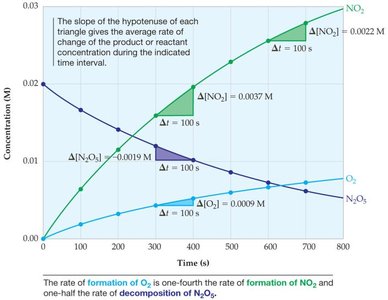
Relating Rates of Reactants and Products
For a general reaction: , the rate can be expressed as:
This ensures that the rate is consistent regardless of which species is monitored.
Rate Laws and Reaction Order
Definition of Rate Law
The rate law expresses the relationship between the reaction rate and the concentrations of reactants. For a reaction :
k: Rate constant (depends on temperature and reaction pathway)
m, n: Reaction orders with respect to A and B (determined experimentally)
Overall Order:
Determining Rate Laws
Rate laws must be determined experimentally, often using the method of initial rates. By varying the concentrations of reactants and measuring the initial rate, the order with respect to each reactant can be found.
Example: For the reaction , if the rate law is , tripling [Cl_2] triples the rate, while quadrupling [NO] increases the rate by a factor of 16.
Units of the Rate Constant (k)
The units of k depend on the overall order of the reaction:
Overall Order | Units of k |
|---|---|
Zero | M/s |
First | 1/s |
Second | 1/(M·s) |
Third | 1/(M2·s) |

Integrated Rate Laws
Zero, First, and Second Order Reactions
Integrated rate laws relate the concentration of reactants to time and allow calculation of concentrations at any point during the reaction.
Order | Integrated Rate Law | Linear Plot | Half-life |
|---|---|---|---|
Zero | vs. | (not constant) | |
First | vs. | (constant) | |
Second | vs. | (not constant) |

Graphical Determination of Reaction Order
To determine the order of a reaction, plot the appropriate function of concentration versus time:
Zero Order: vs. is linear
First Order: vs. is linear
Second Order: vs. is linear

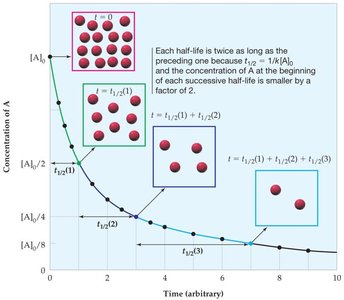
Half-Life of a Reaction
The half-life () is the time required for the concentration of a reactant to decrease by half. For first-order reactions, the half-life is constant and given by:
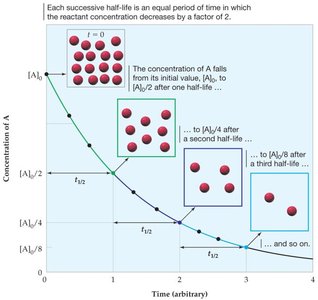
Zero Order: depends on initial concentration.
Second Order: depends on both k and initial concentration.
Applications of Integrated Rate Laws
Drug Metabolism Example
First-order kinetics are common in drug metabolism. For example, the anti-tumor drug cisplatin follows first-order decay in the bloodstream. The concentration at time t can be calculated using:
Environmental Chemistry Example
Many pesticides degrade via first-order kinetics. The time to reach a safe concentration can be calculated using the integrated rate law and the known half-life.
Second Order Reactions
Integrated Rate Law and Half-Life
For a second-order reaction, the integrated rate law is:
The half-life is not constant and is given by:
Reaction Rates and Temperature: The Arrhenius Equation
Collision Theory and Temperature Effects
According to collision theory, molecules must collide with sufficient energy and proper orientation to react. Increasing temperature increases both the frequency and energy of collisions, thus increasing the reaction rate.
Arrhenius Equation: Relates the rate constant k to temperature and activation energy.
A: Frequency factor (related to collision frequency and orientation)
E_a: Activation energy
R: Gas constant
T: Temperature in Kelvin
By plotting vs. , the activation energy can be determined from the slope.
Additional info: These notes cover the core concepts of chemical kinetics, including reaction rates, rate laws, integrated rate laws, and the effect of temperature on reaction rates, as outlined in a typical general chemistry curriculum.
