 Back
BackChemical Kinetics: Temperature, Reaction Mechanisms, and Catalysis
Study Guide - Smart Notes

Chemical Kinetics: Effect of Temperature and Reaction Mechanisms
Temperature Dependence of Reaction Rate
The rate of a chemical reaction depends not only on the concentration of reactants but also on temperature. The rate law is typically expressed as:
Rate Law:
Rate Constant (k): The value of k changes with temperature, and its dependence is described by the Arrhenius Equation.
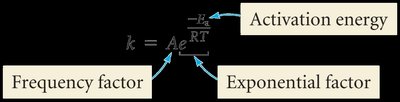
The Arrhenius Equation is:
Where: A = Frequency factor = Activation energy R = Gas constant (8.314 J mol–1 K–1) T = Temperature (K)
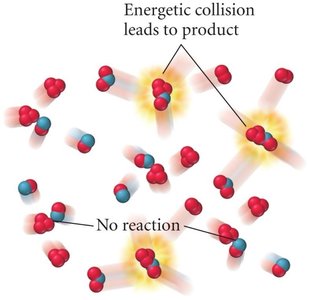
Collision Theory
Collision theory explains why and how reactions occur, especially in the gas phase. A reaction only takes place after a collision between reactant particles. However, not all collisions result in a reaction.
Successful Collisions: Must have sufficient energy to break bonds and proper orientation for new bonds to form.
Frequency Factor (A): Represents the number of approaches to the activation barrier per unit time.
Exponential Factor: Fraction of molecules with enough energy to overcome the activation barrier, depends on temperature.

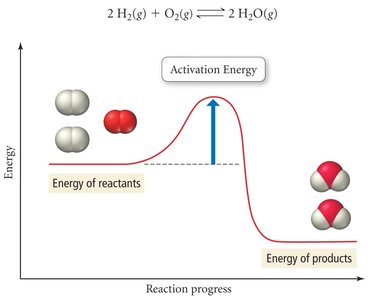
Activation Energy and Potential Energy Diagrams
Activation energy () is the minimum energy required for a reaction to occur. It is the energy difference between reactants and the transition state (activated complex).
High : Slower reaction
Low : Faster reaction
Transition State: High-energy, unstable arrangement of atoms between reactants and products
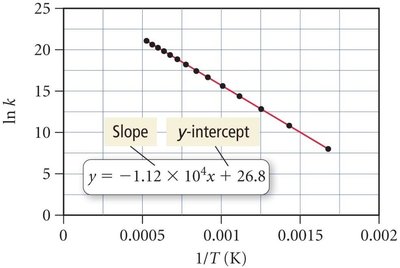
Arrhenius Plots
The Arrhenius equation can be rearranged to a linear form for graphical analysis:
Plotting vs yields a straight line with slope and y-intercept .
Reaction Mechanisms
Elementary Steps and Molecularity
Reactions often proceed through a series of steps called a mechanism. Each step is an elementary step, which cannot be broken down further and proceeds as written.
Molecularity: Number of reactant particles involved in an elementary step (unimolecular, bimolecular, termolecular).
Rate Law for Elementary Steps: Determined directly from the reactants in that step.
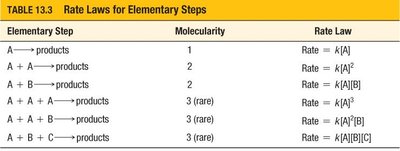
Elementary Step | Molecularity | Rate Law |
|---|---|---|
A → products | 1 | Rate = k[A] |
A + A → products | 2 | Rate = k[A]2 |
A + B → products | 2 | Rate = k[A][B] |
A + A + A → products | 3 (rare) | Rate = k[A]3 |
A + A + B → products | 3 (rare) | Rate = k[A]2[B] |
A + B + C → products | 3 (rare) | Rate = k[A][B][C] |
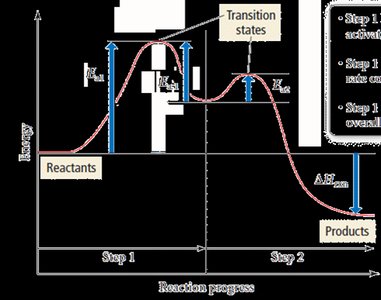
Rate-Determining Step
In a multi-step mechanism, the slowest step is the rate-determining step. The overall rate law for the reaction is governed by this step.
Example: If step 1 is slow and involves NO2, the rate law is Rate = k[NO2]2.

Steady-State Approximation
When intermediates are difficult to measure, the steady-state approximation assumes their concentration remains constant during the reaction. The rate of formation equals the rate of consumption.
Application: Used to express the rate law in terms of measurable reactants and products.

Catalysis
Role of Catalysts
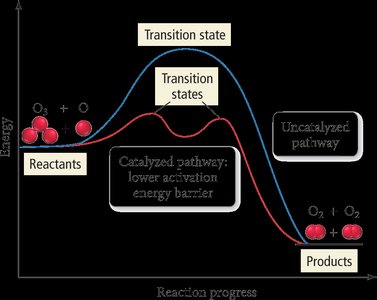
A catalyst provides an alternative reaction pathway with lower activation energy, increasing the reaction rate without being consumed.
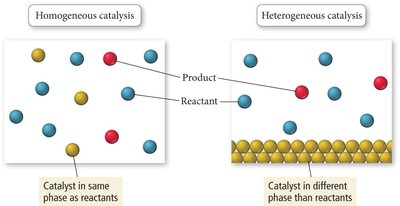
Homogeneous Catalysts: Same phase as reactants
Heterogeneous Catalysts: Different phase than reactants
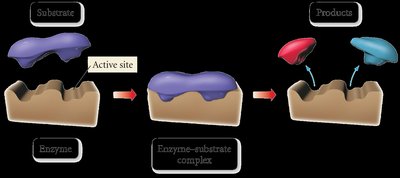
Enzymes: Biological catalysts that orient substrates for reaction
Summary Table: Key Equations
Equation | Description |
|---|---|
Arrhenius equation for rate constant | |
Linearized Arrhenius equation | |
Rate = k[Reactant]n | General rate law |
Rate = k[NO2]2 | Example rate law for a bimolecular step |
