 Back
BackChemical Quantities and Aqueous Reactions: Stoichiometry, Limiting Reactants, and Yield
Study Guide - Smart Notes

Writing and Balancing Chemical Equations
Definitions and Representation
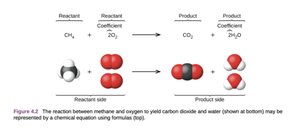
Chemical equations are symbolic representations of chemical reactions, showing the reactants and products, their physical states, and the stoichiometric coefficients. The reactants are listed on the left side, and the products on the right. States of matter are indicated as (s) for solid, (l) for liquid, (g) for gas, and (aq) for aqueous solution.
Chemical equation: A concise way to represent a chemical reaction using chemical formulas and symbols.
Reactants: Substances consumed during the reaction (left side).
Products: Substances formed during the reaction (right side).
Coefficients: Numbers placed before formulas to indicate the number of units involved.
Balancing Chemical Equations
Balancing chemical equations ensures the law of conservation of mass is obeyed: matter is neither created nor destroyed in a chemical reaction. Only coefficients are changed to balance equations; subscripts in chemical formulas must never be altered.
Law of Conservation of Mass: The total mass of reactants equals the total mass of products.
Balance more complex formulas first, treat polyatomic ions as units, and balance free elements last.
Remove fractional coefficients by multiplying all coefficients by the denominator if necessary.
Check that the number of each type of atom is equal on both sides of the equation.
Stoichiometry: Quantitative Relationships in Chemical Reactions
Definition and Importance
Stoichiometry is the study of the quantitative relationships between the amounts of reactants and products in a chemical reaction. It allows chemists to predict how much product will form from given reactants or how much reactant is needed to produce a desired amount of product.
Stoichiometric coefficients indicate the fixed ratios of reactants and products (in moles, not mass).
Example: In the reaction , 2 moles of sodium azide produce 2 moles of sodium and 3 moles of nitrogen gas.
Everyday Analogy
Stoichiometry can be compared to following a recipe, where specific amounts of ingredients yield a certain amount of product (e.g., cookies).


Stoichiometric Calculations
Use molar masses to convert between grams and moles.
Use coefficients from the balanced equation to set up mole ratios for conversions.
Limiting Reactant and Excess Reactant
Definitions
Limiting Reactant: The reactant that is completely consumed first, thus limiting the amount of product formed.
Excess Reactant: The reactant that remains after the reaction is complete.
Theoretical Yield: The maximum amount of product that can be formed from the limiting reactant.
The limiting reactant determines the maximum amount of product that can be made, while the excess reactant is left over after the reaction.

Identifying the Limiting Reactant
Calculate the amount of product that can be formed from each reactant.
The reactant that produces the least amount of product is the limiting reactant.
Use the limiting reactant to determine the amounts of all products and leftover reactants.
Example: In the reaction , if 0.588 mol of nitrogen and 0.632 mol of hydrogen are mixed, calculate the amount of ammonia produced and identify the limiting reactant.
Theoretical Yield, Actual Yield, and Percent Yield
Definitions and Calculations
Theoretical Yield: The calculated maximum amount of product possible from the limiting reactant.
Actual Yield: The amount of product actually obtained from the reaction (usually less than theoretical yield due to losses).
Percent Yield: A measure of reaction efficiency, calculated as:

Calculating Excess Reactant
To find the leftover excess reactant: Leftover amount = initial amount – reacted amount.
Use stoichiometry to determine how much of the excess reactant was consumed.
Summary Table: Key Stoichiometric Terms
Term | Definition |
|---|---|
Limiting Reactant | Reactant that determines the maximum amount of product formed |
Excess Reactant | Reactant that remains after the reaction is complete |
Theoretical Yield | Maximum possible amount of product |
Actual Yield | Amount of product actually obtained |
Percent Yield | Efficiency of reaction, calculated as |
Practice Problems
Balance the following equation:
Given 10.0 g of sucrose and 10.0 g of oxygen, determine the limiting reactant and theoretical yield of water for the reaction
If 4.32 g of water are produced in the above reaction, calculate the percent yield.
