 Back
BackChemical Quantities and Aqueous Reactions: Structured Study Notes
Study Guide - Smart Notes

CHAPTER 4: Chemical Quantities and Aqueous Reactions
Writing and Balancing Chemical Equations
Chemical equations are symbolic representations of chemical reactions, showing the reactants and products, their physical states, and the quantitative relationships between them. Balancing chemical equations is essential to obey the Law of Conservation of Mass, ensuring the same number of each atom type on both sides of the equation.
Chemical Reaction: A process in which substances are converted into different substances by rearrangement and exchange of atoms.
Chemical Equation: Describes the chemical change using formulas and symbols.
Balancing Steps:
Take inventory of elements and their numbers on each side.
Balance atoms in complex substances first, then pure elements.
Balance free elements last, adjusting coefficients as needed.
Clear fractional coefficients by multiplying the entire equation.
Check that the equation is balanced for all atom types.
Example:



States of Reactants and Products
Physical states are indicated in chemical equations using abbreviations:
Abbreviation | State |
|---|---|
(g) | Gas |
(l) | Liquid |
(s) | Solid |
(aq) | Aqueous (water solution) |

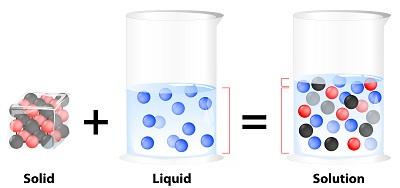
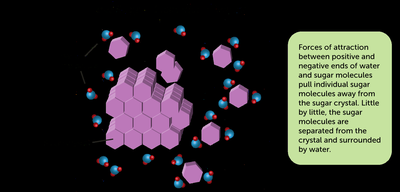
Solutions and Solubility
A solution is a homogeneous mixture of two substances, consisting of a solute (minority component) and a solvent (majority component). An aqueous solution is one where water is the solvent.
Solute: Component that changes state or is present in lesser amount.
Solvent: Component that retains its state or is present in greater amount.
Aqueous Solution: Water acts as the solvent.

Dissolution: Electrolyte and Nonelectrolyte Solutions
Electrolytes are substances that dissolve in water to form solutions that conduct electricity due to the presence of ions. Nonelectrolytes dissolve without forming ions and do not conduct electricity.
Electrolytes: Ionic compounds and molecular acids that dissociate into ions.
Nonelectrolytes: Most molecular compounds (except acids) that do not dissociate.
Strong Electrolytes: Completely ionize in water.
Weak Electrolytes: Partially ionize in water.

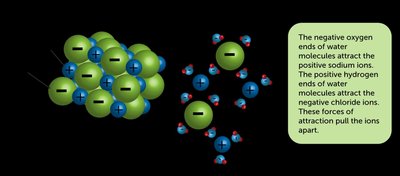
Solubility of Ionic Compounds
Solubility rules help predict whether an ionic compound will dissolve in water. These rules are hierarchical and must be applied in order.
Rule | Description |
|---|---|
1 | All salts containing group 1 cations and ammonium ions are soluble. |
2 | All nitrates, acetates, chlorates, and perchlorates are soluble. |
3 | Salts containing Ag+, Pb2+, and Hg2+ are insoluble. |
4 | Most chlorides, bromides, and iodides are soluble. |
5 | Sulfates are soluble except those containing Ca2+, Sr2+, Ba2+. |
6 | Carbonates, hydroxides, oxides, phosphates, and sulfides are generally insoluble. |
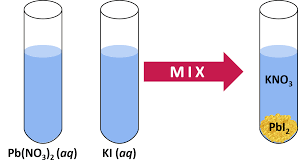
Precipitation Reactions
Precipitation reactions occur when two aqueous solutions of ionic compounds are mixed and produce an insoluble product (precipitate).
Precipitate: The insoluble product formed.
Example:

Writing Chemical Equations for Precipitation Reactions
To write equations for precipitation reactions, follow these steps:
Write formulas for reactants.
Identify ions present and possible products.
Use solubility rules to determine insoluble products.
If a product is insoluble, write the net ionic equation by canceling spectator ions.


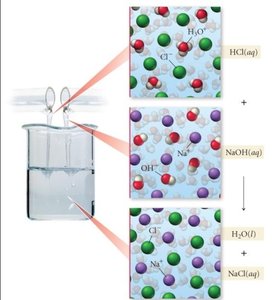
Acid-Base (Neutralization) Reactions
Acid-base reactions involve an acid reacting with a base to produce water and a salt. According to the Arrhenius definition, acids produce H+ ions and bases produce OH- ions in aqueous solution.
Acid: Produces H+ ions in water.
Base: Produces OH- ions in water.
Neutralization: H+ from acid combines with OH- from base to form water.
Example:


Gas-Evolution Reactions
Gas-evolution reactions occur when two aqueous solutions mix to form a gaseous product that bubbles out of solution. Some reactions form gases directly, while others form intermediates that decompose to gases.
Reactant Type | Intermediate Product | Gas Evolved | Example |
|---|---|---|---|
Sulfides | None | H2S | 2 HCl (aq) + K2S (aq) → H2S (g) + 2 KCl (aq) |
Carbonates/Bicarbonates | H2CO3 | CO2 | 2 HCl (aq) + Na2CO3 (aq) → CO2 (g) + H2O (l) + 2 NaCl (aq) |
Sulfites/Bisulfites | H2SO3 | SO2 | 2 HCl (aq) + K2SO3 (aq) → SO2 (g) + H2O (l) + 2 KCl (aq) |
Ammonium | NH4OH | NH3 | NH4Cl (aq) + KOH (aq) → NH3 (g) + H2O (l) + KCl (aq) |

Oxidation-Reduction (Redox) Reactions
Redox reactions involve the transfer of electrons between reactants. Oxidation is the loss of electrons, and reduction is the gain of electrons. The oxidation state (number) helps track electron transfer.
Oxidation: Loss of electrons; increase in oxidation state.
Reduction: Gain of electrons; decrease in oxidation state.
Oxidizing Agent: Causes oxidation; is reduced itself.
Reducing Agent: Causes reduction; is oxidized itself.
Example:
Rules for Assigning Oxidation States
Oxidation states are assigned based on a set of hierarchical rules:
Element: 0
Monoatomic ion: Equal to ion charge
Sum in neutral molecule: 0; in polyatomic ion: equals ion charge
Group 1 metals: +1; Group 2 metals: +2
Hydrogen: Usually +1
Fluorine: Always -1; other Group 17: usually -1; Oxygen: usually -2; Group 16: usually -2; Group 15: usually -3
Balancing Redox Reactions by the Half-Reaction Method
Redox reactions are balanced by separating them into oxidation and reduction half-reactions, balancing for mass and charge, and then combining them.
Assign oxidation states to all atoms.
Separate into two half-reactions.
Balance elements other than H and O.
Balance O by adding H2O; balance H by adding H+.
Balance charge by adding electrons.
Equalize electrons in both half-reactions.
Add half-reactions and verify balance.
Reaction Stoichiometry
Stoichiometry is the numerical relationship between chemical amounts in a balanced equation. It allows prediction of product amounts and determination of reactant requirements.
Coefficients: Specify relative amounts in moles of each substance.
Example:
Stoichiometric Ratios: Used for conversions between reactants and products.
Limiting Reactant, Theoretical and Percent Yield
In reactions with multiple reactants, the limiting reactant is the one completely consumed first, determining the maximum product (theoretical yield). Actual yield is the amount produced, and percent yield is the ratio of actual to theoretical yield.
Theoretical Yield: Maximum possible product based on limiting reactant.
Actual Yield: Amount actually produced.
Percent Yield:
Excess Reactant: Not completely consumed.
Solution Concentration and Solution Stoichiometry
Molarity (M) is the amount of solute (in moles) per liter of solution. It is used to prepare solutions and perform stoichiometric calculations.
Molarity: , where is moles of solute and is volume in liters.
Preparation: Calculate mass of solute needed for desired molarity and volume.
Solution Dilution: (moles of solute remain constant).
Stoichiometry: Use molarity to convert between reactant and product amounts.
Tables and Visuals
Tables are used throughout to summarize solubility rules, acid/base names and formulas, and gas-evolution reaction types. Molecular models and diagrams visually reinforce concepts such as dissolution, precipitation, and acid-base reactions.








