 Back
BackChemical Quantities, Moles, and Chemical Reactions: Core Concepts and Calculations
Study Guide - Smart Notes

Chemical Quantities and the Mole Concept
Formula Weight and Molecular Weight
The formula weight (FW) of a substance is the sum of the atomic weights (AW) of all atoms in its chemical formula. For molecules, this is also called the molecular weight (MW). These values are essential for converting between mass and moles in chemical calculations.
Formula Weight Calculation: Add the atomic weights of each atom in the formula, multiplied by the number of each atom present.
Units: Atomic mass units (amu) are used for individual molecules or formula units.
Example: The formula weight of H2SO4 is calculated as follows:

Percentage Composition from Chemical Formulas
The percentage composition of an element in a compound is the percent by mass of that element in the compound. This is useful for determining empirical formulas and analyzing chemical samples.
Formula:

Example Calculation: For C12H22O11 (sucrose):

The Mole and Avogadro's Number
Definition and Importance
The mole (mol) is the SI unit for the amount of substance. One mole contains exactly 6.022 × 1023 particles (Avogadro's number), whether they are atoms, molecules, or ions. This allows chemists to count particles by weighing them.
1 mole = 6.022 × 1023 particles
Examples:
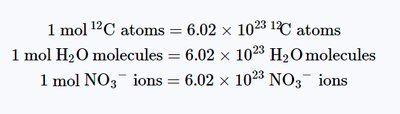
Visualizing the Mole
One mole of any substance contains Avogadro's number of particles, but the mass of one mole varies depending on the substance's molar mass.

Conversions Using Avogadro’s Constant
Avogadro’s constant is used to convert between the number of particles and the number of moles. This is a fundamental skill in chemical calculations.
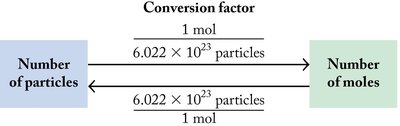
Mole Relationships and Molar Mass
The molar mass is the mass (in grams) of one mole of a substance. It is numerically equal to the formula weight in amu, but the units are grams per mole (g/mol).
Example: Gold (Au) and sulfur trioxide (SO3) have the following relationships:
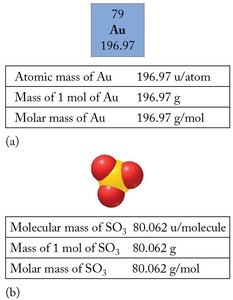
Comparing Mass of a Single Molecule and a Mole
The mass of a single molecule is extremely small compared to the mass of a mole of molecules. For example, one molecule of H2O has a mass of 2.99 × 10−23 g, while one mole of H2O has a mass of 18.0 g.

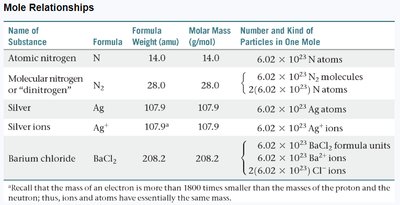
Mole Relationships Table
The following table summarizes the relationships between formula weight, molar mass, and the number of particles in one mole for various substances:
Name of Substance | Formula | Formula Weight (amu) | Molar Mass (g/mol) | Number and Kind of Particles in One Mole |
|---|---|---|---|---|
Atomic nitrogen | N | 14.0 | 14.0 | 6.02 × 1023 N atoms |
Molecular nitrogen | N2 | 28.0 | 28.0 | 6.02 × 1023 N2 molecules 1.20 × 1024 N atoms |
Silver | Ag | 107.9 | 107.9 | 6.02 × 1023 Ag atoms |
Silver ions | Ag+ | 107.9 | 107.9 | 6.02 × 1023 Ag+ ions |
Barium chloride | BaCl2 | 208.2 | 208.2 | 6.02 × 1023 BaCl2 formula units 6.02 × 1023 Ba2+ ions 1.20 × 1024 Cl− ions |
Interconverting Mass, Moles, and Particles
Dimensional Analysis in Chemical Calculations
Dimensional analysis is used to convert between mass, moles, and number of particles. The molar mass and Avogadro’s number serve as conversion factors.
Example: Calculating moles of glucose in a 5.380 g sample:
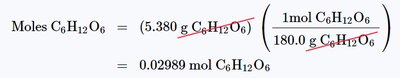
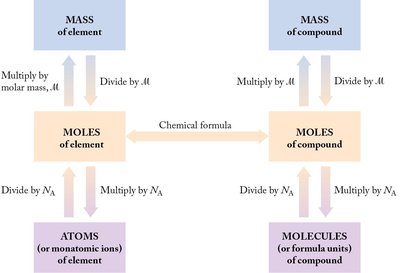
Comprehensive Conversion Map
The following diagram summarizes the relationships and conversions among mass, moles, and particles for both elements and compounds:
Applications: Chemistry and Life
Glucose Levels in Blood
After digestion, glucose is delivered to cells via the blood. Normal blood glucose levels are 70–120 mg/dL, and levels at or above 126 mg/dL are a cause for concern. These values can be converted to grams per deciliter (g/dL) and moles per deciliter (mol/dL) for clinical analysis.
Chemical Equations and Reaction Types
Writing and Balancing Chemical Equations
Chemical equations represent chemical reactions, showing reactants and products with their respective coefficients. Balancing equations ensures the law of conservation of mass is obeyed—there must be the same number of each type of atom on both sides of the equation.
Example: The reaction of hydrogen and oxygen to form water:
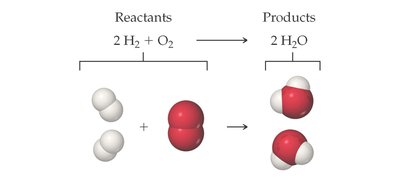
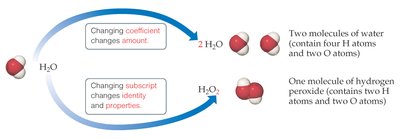
Subscripts vs. Coefficients: Changing a coefficient changes the amount; changing a subscript changes the identity and properties of the substance.
Step-by-Step Balancing Example
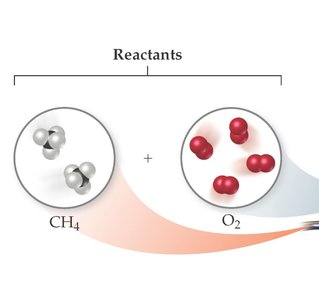
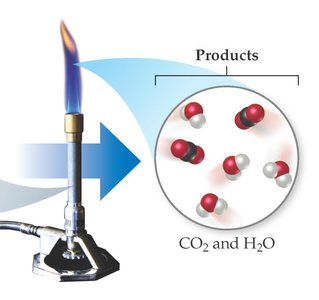
Balancing equations involves adjusting coefficients to ensure equal numbers of each atom on both sides. For example, the combustion of methane (CH4):
Reactants: CH4 and O2
Products: CO2 and H2O
Types of Chemical Reactions
There are five basic types of chemical reactions commonly encountered in general chemistry:
Combination
Decomposition
Single-replacement
Double-replacement
Combustion
Understanding these types helps in predicting products and balancing equations.
