 Back
BackChemical Reactions and Chemical Quantities: Balancing, Stoichiometry, and Environmental Context
Study Guide - Smart Notes

Chapter 4: Chemical Reactions and Chemical Quantities
Introduction to Chemical Reactions
Chemical reactions are processes in which substances (reactants) undergo chemical changes to form new substances (products). Understanding these reactions is fundamental to chemistry, as it allows us to predict the outcomes and quantities involved in chemical processes.
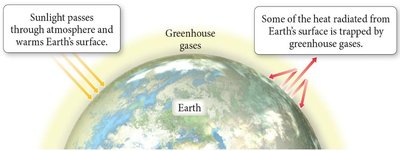
Greenhouse Gases and the Greenhouse Effect
Greenhouse gases in Earth's atmosphere, such as CO2, CH4, and H2O vapor, play a crucial role in regulating Earth's temperature. They allow sunlight to enter the atmosphere and warm the surface, while trapping some of the heat radiated from Earth's surface, preventing it from escaping into space. This balance determines Earth's average temperature.
Key Greenhouse Gases: N2 (78%), O2 (21%), H2O vapor, CO2, CH4, NO2, O3, CFCs
Greenhouse Effect: Essential for maintaining habitable temperatures but excessive greenhouse gases can lead to global warming.
Global Warming: Trends and Causes
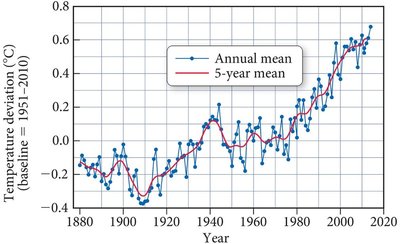
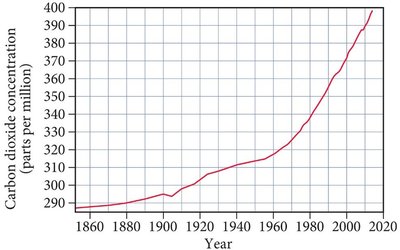
Scientific measurements show a rise in atmospheric temperature and CO2 levels since the late 19th century. The correlation between these trends suggests a causal relationship, largely attributed to human activities such as fossil fuel combustion.
Temperature Rise: Average increase of 0.7°C since 1860.
CO2 Increase: 38% rise in atmospheric CO2 levels over the same period.
Sources of Increased CO2
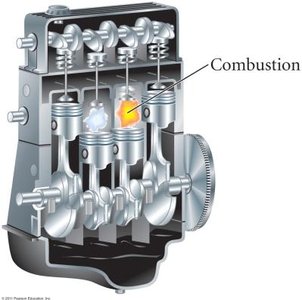
CO2 is produced from both natural and anthropogenic sources. The combustion of fossil fuels is a major contributor, while volcanic activity also emits CO2 but to a much lesser extent.
Combustion Reaction Example:
Volcanic Action: Natural source, but less significant compared to fossil fuel combustion.

Chemical Equations and Balancing
Writing Chemical Equations
Chemical equations represent the reactants and products involved in a reaction, including their physical states. The equation provides information about the formulas, states, and relative quantities of substances.
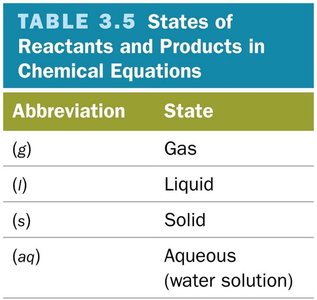
States of Matter: (g) = gas, (l) = liquid, (s) = solid, (aq) = aqueous solution
Abbreviation | State |
|---|---|
(g) | Gas |
(l) | Liquid |
(s) | Solid |
(aq) | Aqueous (water solution) |
Balancing Chemical Equations
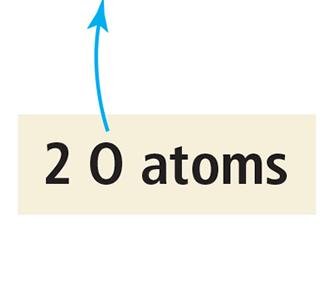
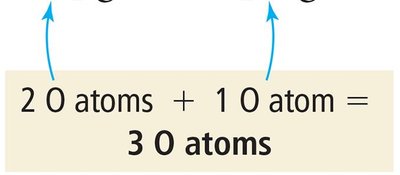
Balancing chemical equations ensures the law of conservation of mass is upheld, meaning the number of atoms of each element is the same on both sides of the equation. This is achieved by adjusting coefficients, not subscripts.
Steps to Balance:
Write the skeletal equation.
Balance atoms in complex substances first.
Balance free elements last.
Clear fractional coefficients by multiplying all coefficients by the denominator.
Check atom balance on both sides.
Example:
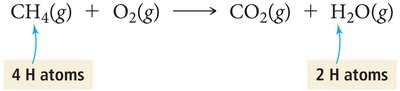

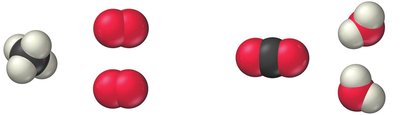
Balancing Equations with Polyatomic Ions
When polyatomic ions appear unchanged on both sides of the equation, they can be balanced as units. This simplifies the balancing process for reactions involving compounds like strontium chloride and lithium phosphate.
Example:
Balance cations first, then anions, and check the total number of ions.
Stoichiometry: Quantitative Relationships in Reactions
Definition and Importance
Stoichiometry is the study of the numerical relationships between the quantities of reactants and products in a balanced chemical reaction. It allows chemists to predict the amounts of products formed and the reactants required for a given reaction.
Enables calculation of product yield and reactant consumption.
Based on the coefficients in balanced equations.









Stoichiometry Analogy: Recipes
Stoichiometry can be compared to recipes, where the relationship between ingredients determines the number of products (e.g., pancakes) that can be made. This analogy helps visualize how reactant quantities affect product yield.
Example: 2 eggs + 1 cup flour + 1/2 tsp baking powder = 5 pancakes
Scaling up: 8 eggs yields 20 pancakes






Mole-to-Mole Conversions
The coefficients in a balanced chemical equation specify the relative amounts in moles of each substance involved. These ratios are used to convert between moles of reactants and products.
Example:
2 mol C8H18 : 25 mol O2 : 16 mol CO2 : 18 mol H2O

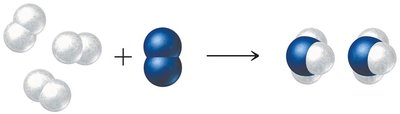
Example: Ammonia Synthesis (Haber Process)
In the Haber process, nitrogen reacts with hydrogen to form ammonia. The balanced equation provides the mole ratios needed for stoichiometric calculations.
Equation:
3 mol H2 : 1 mol N2 : 2 mol NH3
Example calculation: 3 mol N2 × (2 mol NH3 / 1 mol N2) = 6 mol NH3


Mass-to-Mass Conversions
Stoichiometry also allows conversion between masses of reactants and products using molar masses and mole ratios from the balanced equation.
General Plan:
Convert mass of A to moles of A using molar mass.
Use mole ratio to convert moles of A to moles of B.
Convert moles of B to mass of B using molar mass.
Example:
5.0 × 102 g C8H18 produces 1.5 × 103 g CO2





Environmental Application: CO2 Production
Stoichiometric calculations are used to estimate the mass of CO2 produced from global gasoline combustion, highlighting the environmental impact of chemical reactions.
Combustion of 4.0 × 1015 g octane produces 1.2 × 1016 g CO2
Volcanoes emit less than 2% of the CO2 produced by fossil fuel combustion annually.










Summary Table: Key Stoichiometric Relationships
Type of Conversion | Equation/Process | Example |
|---|---|---|
Mole-to-Mole | 2 mol C8H18 : 16 mol CO2 | |
Mass-to-Mass | Use molar mass and mole ratio | 5.0 × 102 g C8H18 → 1.5 × 103 g CO2 |
Environmental Impact | Global CO2 production | 1.2 × 1016 g CO2 from gasoline |
Practice Problems and Applications
Balance chemical equations for various reactions.
Calculate moles and masses of reactants and products.
Apply stoichiometry to real-world environmental issues.
Additional info: The notes include environmental context (global warming, CO2 sources), practical analogies (recipes), and stepwise examples for balancing equations and stoichiometric calculations, making them suitable for exam preparation and foundational understanding in general chemistry.
