 Back
BackChemical Reactions and Stoichiometry: Study Guide
Study Guide - Smart Notes

Chemical Reactions and Stoichiometry
What is a Chemical Reaction?
A chemical reaction is a process in which substances (reactants) are transformed into new substances (products) through the breaking and forming of chemical bonds. Chemical reactions are fundamental to chemistry and are classified into several common types based on their patterns and outcomes.
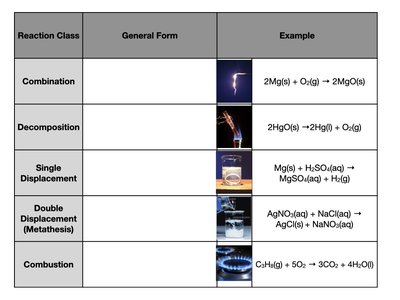
Combination (Synthesis): Two or more substances combine to form a single product.
Decomposition: A single compound breaks down into two or more simpler substances.
Single Displacement: An element replaces another element in a compound.
Double Displacement (Metathesis): Two compounds exchange ions to form two new compounds.
Combustion: A substance reacts with oxygen, releasing energy and producing oxides.
Example Table:
Reaction Class | General Form | Example |
|---|---|---|
Combination | 2Mg(s) + O2(g) → 2MgO(s) | |
Decomposition | 2HgO(s) → 2Hg(l) + O2(g) | |
Single Displacement | Mg(s) + H2SO4(aq) → MgSO4(aq) + H2(g) | |
Double Displacement (Metathesis) | AgNO3(aq) + NaCl(aq) → AgCl(s) + NaNO3(aq) | |
Combustion | C2H6(g) + 5O2 → 3CO2 + 4H2O(l) |
Writing Chemical Equations
Chemical equations represent chemical reactions using symbols and formulas. Reactants are written on the left, products on the right, separated by an arrow (→). Each substance is accompanied by its physical state and, if necessary, reaction conditions.
Reactants: Substances consumed in the reaction (left side).
Products: Substances formed in the reaction (right side).
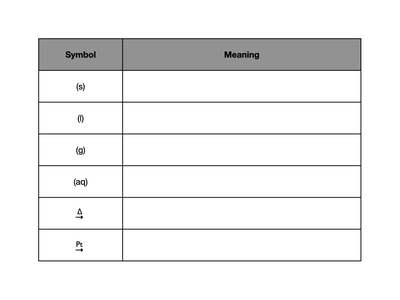
Physical States: Indicated by symbols: (s) for solid, (l) for liquid, (g) for gas, (aq) for aqueous.
Reaction Conditions: Shown above or below the arrow (e.g., Δ for heat, Pt for catalyst).
Common Symbols Table:
Symbol | Meaning |
|---|---|
(s) | Solid |
(l) | Liquid |
(g) | Gas |
(aq) | Aqueous (dissolved in water) |
Δ | Heat is applied |
Pt | Platinum catalyst |
Balancing Chemical Equations
Balancing a chemical equation ensures the Law of Conservation of Mass is obeyed: the number of atoms of each element and the total mass must be the same on both sides of the equation. Coefficients are adjusted to achieve balance, but subscripts (which define the identity of compounds) must not be changed.
Start with an unbalanced equation.
Balance atoms in compounds before those in free elements.
Balance polyatomic ions as units when possible.
Do not alter subscripts.
Example: Balancing the reaction of iron(III) oxide with hydrogen:
Stoichiometry: Relating Amounts in Reactions
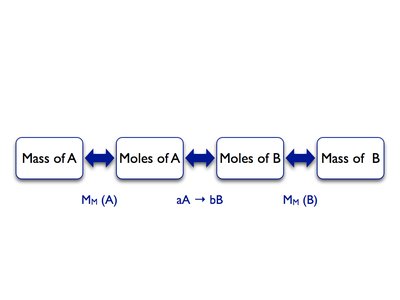
Stoichiometry is the quantitative relationship between reactants and products in a chemical reaction. The coefficients in a balanced equation indicate the ratios of moles, particles, or volumes (for gases) involved.
Mole Ratio: Derived from the coefficients in the balanced equation.
Mass-Mole Calculations: Use molar mass to convert between mass and moles.
Stoichiometric Calculation Flow:
Limiting and Excess Reactants
In reactions with more than one reactant, the limiting reactant is the one that is completely consumed first, thus determining the maximum amount of product formed. The excess reactant is the one left over after the reaction.
Calculate the amount of product each reactant can produce.
The reactant producing the least product is the limiting reactant.
Excess reactant can be calculated by subtracting the amount used from the initial amount.
Example: For the reaction , determine the limiting reactant given masses of Mg and O2.
Theoretical, Actual, and Percent Yield
Yield calculations are important for evaluating reaction efficiency.
Theoretical Yield: The maximum amount of product predicted by stoichiometry.
Actual Yield: The amount of product actually obtained from the experiment.
Percent Yield: The ratio of actual yield to theoretical yield, expressed as a percentage.
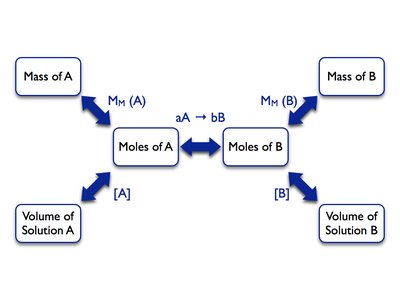
Stoichiometry in Solution Reactions
Stoichiometric calculations can also be applied to reactions occurring in solution, using molarity (M) to relate volume and moles.
Molarity (M): Moles of solute per liter of solution.
Use balanced equations to relate moles of reactants and products.
Convert between mass, moles, and volume as needed.
Gravimetric Analysis
Gravimetric analysis is a quantitative technique where the amount of an analyte is determined by measuring the mass of a product formed.
React sample with a reagent to form a precipitate.
Collect, dry, and weigh the precipitate.
Calculate the mass percent of the analyte in the original sample.
Example: Determining the mass percent of calcium in limestone by precipitating CaC2O4.
Titration and Stoichiometry
Titration is a technique for determining the concentration of a solution by reacting it with a solution of known concentration. The equivalence point is when stoichiometrically equivalent amounts of reactants have reacted. An indicator is used to signal the endpoint of the titration.
Calculate moles of titrant used.
Use balanced equation to relate moles of analyte and titrant.
Determine concentration or percent composition as needed.
Example: Titrating hydrogen peroxide with potassium permanganate to determine percent by mass.
Additional info: Academic context and examples were expanded for completeness and clarity. Tables and images were included only when directly relevant to the explanation.
