 Back
BackChemical Structure and Acid-Base Behavior: Predicting Strength from Molecular Structure
Study Guide - Smart Notes

Ch. 16: Chemical Structure and Acid-Base Behavior
Introduction to Acid-Base Strength and Molecular Structure
The strength of acids and bases is fundamentally linked to their molecular structure. Understanding how atomic and electronic factors influence acid and base behavior allows chemists to predict reactivity and properties of compounds. This section explores the structural determinants of acid and base strength, focusing on binary acids, oxyacids, and amines.
Factors Affecting Acid Strength
Acid strength depends on how easily a molecule can donate a proton (H+). The weaker the bond between the acidic proton and the rest of the molecule, the stronger the acid. Several structural factors contribute to this:
Bond Strength: The strength of the H–X bond (where X is the atom bonded to hydrogen) is crucial. Weaker bonds facilitate proton release.
Electronegativity: Highly electronegative atoms stabilize the negative charge on the conjugate base, increasing acid strength.
Atomic Size: Larger atoms form longer, weaker H–X bonds, making acids stronger as you move down a group.
Trends in Acid Strength Across the Periodic Table
Acid strength varies systematically across periods and groups in the periodic table. The following table summarizes these trends for common hydrides:
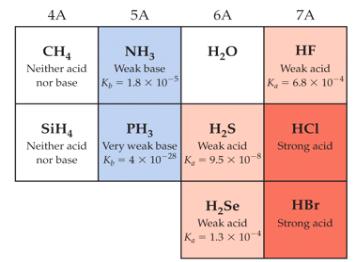
Across a Period: Acid strength increases as the electronegativity of X increases (from left to right).
Down a Group: Acid strength increases as the size of X increases (from top to bottom).
Examples: HF is a weak acid, HCl and HBr are strong acids; NH3 is a weak base, PH3 is a very weak base.
Binary Acids: Structure and Strength
Binary acids consist of hydrogen and one other element (e.g., HCl, HF). Their strength is determined by the bond strength and the ability of the conjugate base to stabilize the negative charge.
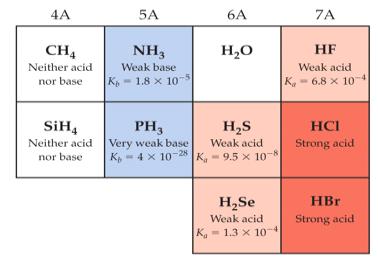
Bond Length: Longer bonds (with larger atoms) are weaker, making the acid stronger.
Electronegativity: Higher electronegativity increases acid strength across a period.
Oxyacids: Structure and Acid Strength
Oxyacids contain hydrogen, oxygen, and another element (the central atom). Their acid strength depends on the electronegativity of the central atom and the number of oxygen atoms attached.

Electronegativity of Central Atom: Higher electronegativity stabilizes the conjugate base, increasing acid strength.
Number of Oxygen Atoms: More oxygen atoms increase acid strength by stabilizing the negative charge via resonance.
Example Table:
Substance | Y—OH | Electronegativity of Y | Dissociation constant |
|---|---|---|---|
Hypochlorous acid | Cl—OH | 3.0 | Ka = 3.0 × 10−8 |
Hypobromous acid | Br—OH | 2.8 | Ka = 2.5 × 10−9 |
Hypoiodous acid | I—OH | 2.5 | Ka = 2.3 × 10−11 |
Water | H—OH | 2.1 | Kw = 1.0 × 10−14 |
Acid Strength in Series of Oxyacids
When comparing oxyacids with the same central atom but different numbers of oxygen atoms, acid strength increases with more oxygen atoms. This is due to increased resonance stabilization of the conjugate base.
Example: HNO3 (nitric acid) is stronger than HNO2 (nitrous acid).
Conjugate Bases and Resonance Stabilization
The stability of the conjugate base is a key factor in acid strength. Resonance stabilization, where the negative charge is delocalized over several atoms, increases the strength of the parent acid.
Example: Formic acid and its conjugate base, the formate ion, are stabilized by resonance.
Structure and Basicity of Amines
Amines are common weak bases, structurally related to ammonia. Their basicity is determined by the presence of a lone pair of electrons on the nitrogen atom, which can accept a proton.
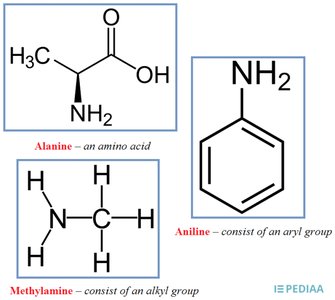
Common Structural Element: All amines have a nitrogen atom with a lone pair.
Basic Behavior: The lone pair allows amines to act as proton acceptors (Brønsted bases).
Types of Amines: Alkyl amines (e.g., methylamine), aryl amines (e.g., aniline), and amino acids (e.g., alanine).
Predicting Conjugate Base Behavior from Acid Structure
The behavior of a conjugate base can be predicted from the structure and acid strength of its parent acid. The more stable the conjugate acid, the weaker the base.

Relative Stability: The stability of the conjugate acid determines the base strength (Kb).
Example: Among CH3NH2, NH2NH2, HONH2, and ClNH2, the base with the most stable conjugate acid has the largest Kb.
