 Back
BackChemical Thermodynamics: Energy, Enthalpy, Entropy, and Spontaneity
Study Guide - Smart Notes

Chemical Thermodynamics
Introduction to Thermodynamics
Chemical thermodynamics is the study of energy changes and energy transfers that accompany chemical and physical processes. Understanding these concepts is essential for predicting whether reactions will occur spontaneously and for quantifying the energy involved in chemical transformations.
Energy: The capacity to do work. It exists in two main forms: kinetic energy (energy of motion) and potential energy (energy due to position or composition).
Thermodynamic Terms: System (the substances and container involved in the process), Surroundings (everything outside the system), and Universe (system + surroundings).
Types of Systems: Open (can exchange matter and energy), Closed (can exchange energy but not matter), Isolated (cannot exchange matter or energy).
The First Law of Thermodynamics
The First Law states that the total energy of the universe is constant. This is also known as the Law of Conservation of Energy: energy is neither created nor destroyed in chemical reactions and physical changes.
State Functions: Properties that depend only on the state of the system, not the path taken. Examples: T (temperature), P (pressure), V (volume), ΔE (internal energy change), ΔH (enthalpy change), S (entropy).
Change in any state function:
Enthalpy and Calorimetry
Enthalpy Change (ΔH)
Enthalpy change, ΔH, is the heat transferred into or out of a system at constant pressure. Most chemical reactions in the lab occur at constant pressure.
(heat at constant pressure)
Exothermic reactions: (heat released)
Endothermic reactions: (heat absorbed)
Calorimetry: Measuring Heat Changes
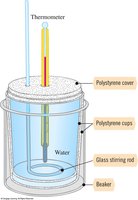
Calorimetry is an experimental technique used to determine the energy change associated with a chemical or physical process. A coffee-cup calorimeter is commonly used for reactions in solution at constant pressure.
Specific heat (s): Amount of heat required to raise the temperature of 1 g of a substance by 1°C.
Heat capacity: Amount of heat gained by the calorimeter.
Heat equation:

Thermochemical Equations and Standard Enthalpy
Thermochemical Equations
Thermochemical equations are balanced chemical reactions with the ΔH value included. They indicate whether a reaction is exothermic or endothermic.
designates an exothermic reaction.
designates an endothermic reaction.
Standard Molar Enthalpy of Formation (ΔHof)
The standard molar enthalpy of formation is the enthalpy change for the reaction in which one mole of a substance is formed from its elements in their most stable forms at 298.15 K and 1.000 atm. The symbol is ΔHof.
Standard molar enthalpies of elements in their most stable forms are zero.
Hess's Law and Bond Energies
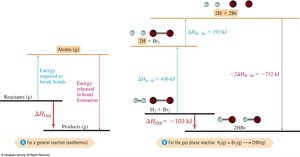
Hess's Law
Hess's Law states that the enthalpy change for a reaction is the same whether it occurs in one step or multiple steps. This is because ΔH is a state function.
Bond Energies
Bond energy is the amount of energy required to break a bond and separate atoms in the gas phase. Breaking bonds always requires energy absorption.
Bond energies can be used to estimate reaction enthalpies.
Internal Energy and Calorimetry at Constant Volume
Internal Energy (ΔE)
Internal energy, E, is the sum of all energy contained within a substance. The First Law of Thermodynamics relates changes in internal energy to heat flow (q) and work (w):
At constant volume,
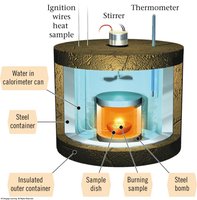
Constant Volume Calorimeter (Bomb Calorimeter)
A bomb calorimeter is used to measure the amount of heat produced or absorbed in a reaction at constant volume. This is a method to measure qv (ΔE) for reactions.



Spontaneity, Entropy, and Free Energy
Spontaneity of Physical and Chemical Changes
Spontaneous changes occur without outside influence and have a natural direction. Nonspontaneous changes require energy input.
Exothermicity and increased disorder both favor spontaneity, but neither alone guarantees it.
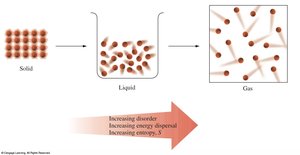
Entropy (S)
Entropy is a measure of disorder or randomness in a system. The Second Law of Thermodynamics states that the universe tends toward increasing entropy in spontaneous changes.
favors spontaneity (disorder increases).
does not favor spontaneity (disorder decreases).
Third Law of Thermodynamics
The Third Law states that the entropy of a pure, perfect, crystalline solid at 0 K is zero. This allows measurement of absolute entropy values.

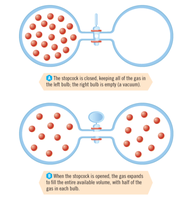
Predictable Changes in Entropy
Entropy increases with temperature.
Entropy increases sharply during phase changes (solid → liquid → gas).
Entropy increases as gas volume increases.
Mixing substances or dissolving solids increases entropy.
Increasing the number of moles of gas increases entropy.


Gibbs Free Energy and Spontaneity
Gibbs Free Energy (ΔG)
Gibbs Free Energy, ΔG, is a state function that combines enthalpy and entropy to predict spontaneity at constant temperature and pressure.
Sign conventions:
: spontaneous process
: system at equilibrium
: nonspontaneous process

Standard Gibbs Free Energy of Formation (ΔGof)
ΔGof is the free energy change when one mole of a compound is formed from its elements in their standard states.
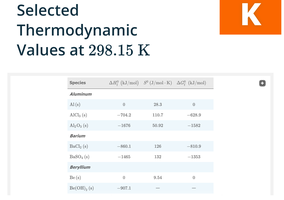
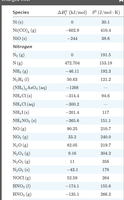
Summary Table: Thermodynamic Values
Thermodynamic values for various substances are tabulated for reference and calculation of enthalpy, entropy, and free energy changes.
Species | ΔHof (kJ/mol) | So (J/mol·K) | ΔGof (kJ/mol) |
|---|---|---|---|
Al(s) | 0 | 28.3 | 0 |
Al2O3(s) | -784.2 | 50.92 | -603.9 |
Ba(s) | 0 | 144 | 0 |
BaO(s) | -553.5 | 70.52 | -513.2 |
Ba(OH)2(s) | -960.1 | 126 | -833.3 |
Be(s) | 0 | 9.44 | 0 |
Be(OH)2(s) | -987.1 | 56.6 | -833.3 |

Key Equations
Heat equation:
Change in state function:
Enthalpy of reaction:
Entropy change:
Gibbs Free Energy:
Additional info: Academic context and examples have been added to clarify concepts and make the notes self-contained for exam preparation.
