 Back
BackChemical Thermodynamics: Energy, Enthalpy, Entropy, and Spontaneity
Study Guide - Smart Notes

Chemical Thermodynamics
Introduction to Thermodynamics
Chemical thermodynamics is the study of energy changes and transfers that occur during chemical and physical processes. It provides a framework for understanding how and why reactions occur, and whether they are energetically favorable.
Energy: The capacity to do work.
Kinetic Energy: Energy of motion.
Potential Energy: Energy due to position or composition.
The First Law of Thermodynamics
Law of Conservation of Energy
The first law states that the total energy of the universe is constant. Energy can neither be created nor destroyed, only transferred or transformed.
Exothermic reactions: Release energy as heat; products have lower potential energy than reactants.
Endothermic reactions: Absorb energy as heat; products have higher potential energy than reactants.
Thermodynamic Terms
System, Surroundings, and Universe
Thermodynamics distinguishes between the system (the part under study) and its surroundings (everything else). The universe is the sum of both.
System: Chemicals and container involved in the process.
Surroundings: Everything outside the system.
Types of Systems: Open, closed, and isolated systems.
Thermodynamic State: Set of conditions specifying all properties of the system.
State Functions
State functions depend only on the state of the system, not the path taken to reach it. Examples include temperature (T), pressure (P), volume (V), internal energy (ΔE), enthalpy (ΔH), and entropy (S).
Change in a state function: $\Delta X = X_{\text{final}} - X_{\text{initial}}$
Enthalpy Change (ΔH)
Definition and Measurement
Enthalpy change, ΔH, is the heat transferred at constant pressure. It is a key quantity in chemical reactions.
$\Delta H = q_p$ (heat at constant pressure)
$\Delta H_{\text{rxn}} = H_{\text{products}} - H_{\text{reactants}}$
If $\Delta H_{\text{rxn}} < 0$, the reaction is exothermic.
If $\Delta H_{\text{rxn}} > 0$, the reaction is endothermic.
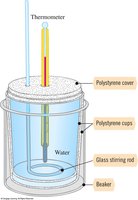
Calorimetry: Measuring Heat Changes
Calorimetry is the experimental technique used to measure energy changes in chemical and physical processes. A coffee-cup calorimeter is commonly used for reactions at constant pressure.
Specific heat (s): Heat required to raise 1 g of a substance by 1°C.
Heat capacity: Amount of heat absorbed by the calorimeter.
Heat equation: $q = m \times s \times \Delta T$

Thermochemical Equations
Balanced Reactions with Enthalpy
Thermochemical equations include both the balanced chemical reaction and the associated ΔH value. They indicate whether a reaction is exothermic or endothermic.
$\Delta H < 0$: Exothermic reaction
$\Delta H > 0$: Endothermic reaction
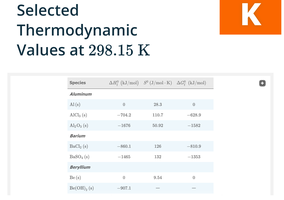
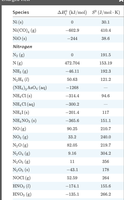
Standard Molar Enthalpies of Formation (ΔHof)
Definition and Application
The standard molar enthalpy of formation is the enthalpy change when one mole of a compound is formed from its elements in their standard states at 298.15 K and 1 atm. For elements in their most stable form, ΔHof = 0.
Hess’s Law
Calculating Enthalpy Changes
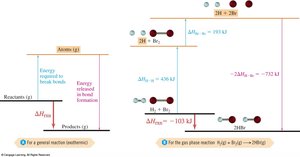
Hess’s Law states that the enthalpy change for a reaction is the same whether it occurs in one step or multiple steps. This is because enthalpy is a state function.
Allows calculation of ΔH for reactions using known enthalpy changes of other reactions.
Bond Energies
Definition and Use
Bond energy is the energy required to break a bond in the gas phase. It is always positive, as breaking bonds requires energy input. Average bond energies can be used to estimate reaction enthalpies.
Example bond energies: F2 = 159 kJ/mol, O2 (double bond) = 498 kJ/mol, N2 (triple bond) = 946 kJ/mol.
Changes in Internal Energy (ΔE)
Internal Energy and the First Law
Internal energy, E, includes all forms of energy within a substance. The first law relates changes in internal energy to heat (q) and work (w):
$\Delta E = q + w$
Relationship of ΔH and ΔE
ΔH and ΔE differ by the amount of expansion work (PΔV) done by the system. At constant pressure and temperature, this difference is usually small unless there is a change in the number of moles of gas.
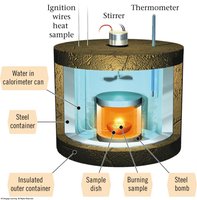
Constant Volume Calorimeter (Bomb Calorimeter)
Measurement of Heat at Constant Volume
A bomb calorimeter is used to measure heat changes at constant volume, providing ΔE for reactions. It is especially useful for combustion reactions.



Spontaneity of Physical and Chemical Changes
Spontaneous vs. Nonspontaneous Changes
Spontaneous changes occur naturally without external influence, while nonspontaneous changes require energy input. Spontaneity depends on both enthalpy and entropy.
Spontaneous examples: Rusting of iron, melting of ice at room temperature.
Nonspontaneous examples: Compressing a gas, freezing ice at room temperature.
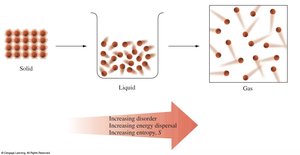
Entropy (S)
Definition and Laws
Entropy is a measure of disorder or randomness. The second law states that the universe tends toward increasing entropy in spontaneous processes. The third law states that the entropy of a perfect crystal at 0 K is zero.
If $\Delta S > 0$, disorder increases (favors spontaneity).
If $\Delta S < 0$, disorder decreases (does not favor spontaneity).

Predictable Changes in Entropy
Entropy increases with phase changes (solid → liquid → gas), temperature, volume, mixing, and molecular complexity.
$S_{\text{gas}} > S_{\text{liquid}} > S_{\text{solid}}$
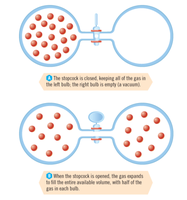
Mixing substances or dissolving solids increases entropy.
Increasing the number of moles of gas increases entropy.


Free Energy Change (ΔG) and Spontaneity
Gibbs Free Energy
Gibbs free energy, ΔG, combines enthalpy and entropy to predict spontaneity at constant temperature and pressure. It is a state function.
$\Delta G = \Delta H - T\Delta S$
If $\Delta G < 0$, the process is spontaneous.
If $\Delta G > 0$, the process is nonspontaneous.
If $\Delta G = 0$, the system is at equilibrium.

Standard Gibbs Free Energy of Formation (ΔGof)
ΔGof is the free energy change when one mole of a compound is formed from its elements in their standard states.
Summary Table: Key Thermodynamic Quantities
Quantity | Symbol | Definition |
|---|---|---|
Enthalpy | ΔH | Heat change at constant pressure |
Internal Energy | ΔE | Total energy change (heat + work) |
Entropy | S | Measure of disorder |
Gibbs Free Energy | ΔG | Maximum useful work obtainable |
Example: The melting of ice at room temperature is spontaneous because it increases entropy and is favored by the combination of enthalpy and entropy at that temperature.
Additional info: Standard thermodynamic values and tables are essential for calculating enthalpy, entropy, and free energy changes for reactions. The use of calorimeters (coffee-cup and bomb) is fundamental in experimental thermochemistry.
