 Back
BackCHM 109 Exam 1 Study Guide: Solutions, Kinetics, and Intermolecular Forces
Study Guide - Smart Notes

Chapter 5: Solutions and Electrolytes
Electrolytes vs Nonelectrolytes
Electrolytes are substances that dissociate into ions when dissolved in water, allowing the solution to conduct electricity. Nonelectrolytes do not dissociate into ions and thus do not conduct electricity.
Electrolytes: Ionic compounds (e.g., NaCl, KBr) and some acids/bases.
Nonelectrolytes: Molecular compounds (e.g., sugar, ethanol).
Conductivity: Only solutions with free ions (electrolytes) carry current.
Dissociation/Ionization Equations for Strong Electrolytes
Strong electrolytes dissociate completely in water. The dissociation equation shows the separation into ions.
Example:
Factors Affecting Solubility
Solubility depends on temperature, pressure (for gases), and the nature of solute and solvent.
Temperature: Solubility of solids usually increases with temperature; gases decrease.
Pressure: Only affects gases; higher pressure increases solubility.
Nature: "Like dissolves like"—polar solutes dissolve in polar solvents.
Solubility Rules in Water
Solubility rules help predict whether a compound will dissolve in water.
Example: AgCl is insoluble; NaCl is soluble.
Common rules: All nitrates, acetates, and most alkali metal salts are soluble.
Double Displacement Reactions
Double displacement reactions involve the exchange of ions between two compounds, often forming a precipitate.
States: Indicate (aq) for aqueous, (s) for solid, (l) for liquid, (g) for gas.
Molecular, Complete Ionic, and Net Ionic Equations
These equations represent reactions at different levels:
Molecular: Shows compounds as intact units.
Complete Ionic: Shows all strong electrolytes as ions.
Net Ionic: Shows only the ions and molecules directly involved in the reaction.
Dilution Equation
Used to calculate the concentration after dilution:
Chapter 12 & 14: Intermolecular Forces and Solution Concentrations
Intermolecular Forces (IMF)
IMFs are forces between molecules affecting boiling points, melting points, and solubility.
Types: Dispersion (London), dipole-dipole, hydrogen bonding, ion-dipole.
Boiling Point: Stronger IMFs lead to higher boiling points.
Concentration Units
Several units are used to express concentration:
Molarity (M):
Molality (m):
Mole Fraction (\chi):
Mole Percent:
Percent by Mass:
ppm:
Concentration Conversions
Convert between units using molar mass, solution volume, and mass as needed.
Unit Prefixes and Conversions
Giga (G):
Mega (M):
Kilo (k):
Centi (c):
Milli (m):
Micro (\mu):
Nano (n):
Henry’s Law
Describes the solubility of gases in liquids:
Trend: Gas solubility increases with pressure, decreases with temperature.
Raoult’s Law
Relates vapor pressure of a solution to the mole fraction of the solvent:
Effect: Adding solute decreases vapor pressure.
Vapor Pressure
Temperature: Higher temperature increases vapor pressure.
IMF Strength: Stronger IMFs decrease vapor pressure.
Boiling Point: Occurs when vapor pressure equals atmospheric pressure.
Freezing Point Depression and Boiling Point Elevation
Van’t Hoff Factor (i): Number of particles produced by dissociation (e.g., NaCl ).
Osmotic Pressure
R: Use
Molar Mass Calculations
Use any concentration equation containing moles to solve for molar mass if grams are given.
Chapter 15: Chemical Kinetics
Kinetics and Rate Definitions
Kinetics: Study of reaction rates.
Rate: Change in concentration per unit time.
Average Rate: Over a time interval.
Instantaneous Rate: At a specific moment.
Rate Expressions
For reaction :
Factors Affecting Rate
Concentration, temperature, presence of catalyst, surface area.
Rate Laws
General form:
Order in reactants: Exponents , .
Overall order: Sum of exponents.
Units of depend on overall order.
Example: is first order in each, second order overall, units: .
Method of Initial Rates
Compare rates with varying initial concentrations to determine order and .
Integrated Rate Laws and Half-Life
0th order:
1st order:
2nd order:
Half-life:
0th:
1st:
2nd:
Graphs of Integrated Rate Laws
0th: vs time, negative slope.
1st: vs time, negative slope.
2nd: vs time, positive slope.
Kinetics and Collision Theory
Reaction rate depends on frequency and energy of collisions.
Graph interpretation: Endothermic vs exothermic, transition states, number of steps.
Arrhenius Equation
Variables: (rate constant), (frequency factor), (activation energy), (gas constant), (temperature in K).
Two-Point Arrhenius Equation
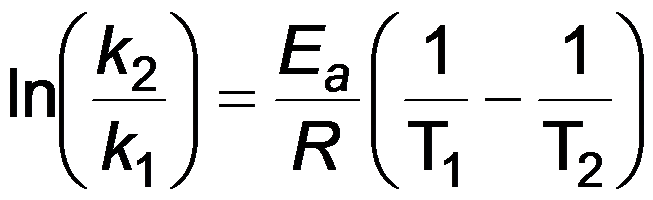
Arrhenius Plot Information
Slope of vs plot is .
Molecularity and Rate Laws
Molecularity: Number of reactant particles in an elementary step.
Rate law for first step: Only include reactants, not intermediates.
Reaction Mechanisms and Catalysts
Mechanism: Sequence of elementary steps.
Catalyst: Lowers activation energy, increases rate, not consumed.
Intermediate: Formed and consumed during reaction.
Types of catalysts: Homogeneous, heterogeneous.
Exam Preparation and Requirements
Study Sources
Textbook, ACS study book, end-of-chapter problems, lecture slides, ALEKS.
Equation Sheet
Provided equations: Gas conversions, rate laws, half-life, Arrhenius equation.
Constants: ,
Exam Logistics
Bring scientific calculator, writing utensil (no red/pink/orange ink).
Show all work for partial credit.
Follow directions for full credit.
Significant figures must be used correctly.
Additional Info
Some questions may come from lecture notes not covered in class.
No practice exams provided; use listed sources for self-study.
