 Back
BackCHM 2046 Final Exam Review – Step-by-Step Study Guidance
Study Guide - Smart Notes

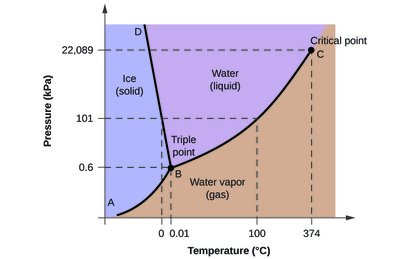
Q4. What phase change occurs from A to B in the phase diagram?
Background
Topic: Phase Changes and Phase Diagrams
This question tests your understanding of how to interpret a phase diagram and identify the phase change that occurs between two points (A and B) on the diagram.
Key Terms and Concepts:
Phase Diagram: A graphical representation showing the states of matter (solid, liquid, gas) of a substance at various temperatures and pressures.
Phase Change: The transition of a substance from one state of matter to another (e.g., melting, freezing, vaporization, condensation, sublimation, deposition).
Triple Point: The unique set of conditions at which all three phases coexist in equilibrium.
Step-by-Step Guidance
Locate points A and B on the phase diagram. Observe which regions (solid, liquid, gas) these points are in.
Determine the direction of the transition (from A to B). Is it moving from the solid region to the gas region, or another combination?
Recall the names of phase changes: - Solid to gas: Sublimation - Solid to liquid: Melting (fusion) - Liquid to gas: Vaporization - Gas to liquid: Condensation - Liquid to solid: Freezing - Gas to solid: Deposition
Match the movement from A to B with the correct phase change based on the regions and direction.
Try solving on your own before revealing the answer!
Final Answer: Sublimation
The movement from point A (solid) to point B (gas) without passing through the liquid phase is called sublimation.
This is a direct transition from solid to gas, which occurs under specific conditions of low pressure and increasing temperature.
Q20. Dinitrogen monoxide decomposes into nitrogen and oxygen when heated. The initial rate of the reaction is 0.450 M/s. What is the rate of change of concentration of N2O?
Background
Topic: Chemical Kinetics – Reaction Rates
This question tests your ability to relate the rate of a reaction to the rate of change in concentration of a reactant, using stoichiometry from the balanced equation.
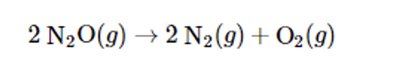
Key Terms and Formulas:
Reaction Rate: The change in concentration of a reactant or product per unit time.
Stoichiometry: The coefficients in the balanced equation relate the rates of consumption and formation of reactants and products.
General Rate Expression:
Step-by-Step Guidance
Write the balanced equation:
Express the rate in terms of the disappearance of N2O:
Set the given rate (0.450 M/s) equal to the rate of formation of N2 or O2 as appropriate, and solve for .
Remember that the rate of disappearance of N2O will be negative, indicating a decrease in concentration.
Try solving on your own before revealing the answer!
Final Answer: -0.900 M/s
Since the rate is given for the formation of N2 (0.450 M/s), and the stoichiometry is 2:2, the rate of disappearance of N2O is twice that value but negative (since it's being consumed).
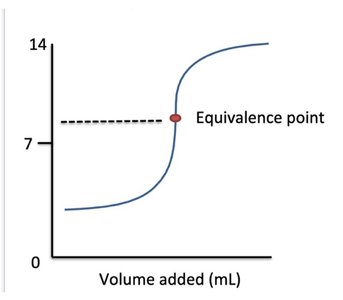
Q39. Describe what is occurring in the following titration curve:
Background
Topic: Acid-Base Titrations and Titration Curves
This question tests your ability to interpret a titration curve and identify the type of titration (strong/weak acid or base) based on the shape and equivalence point.
Key Terms and Concepts:
Titration Curve: A plot of pH versus volume of titrant added during a titration.
Equivalence Point: The point at which the amount of titrant added is stoichiometrically equivalent to the amount of analyte in solution.
Strong Acid/Base vs. Weak Acid/Base: The initial pH and the shape of the curve can help you determine the strength of the acid or base involved.
Step-by-Step Guidance
Observe the starting pH of the curve. Is it above or below 7? This can indicate whether you start with an acid or a base.
Look at the pH at the equivalence point. Is it above, below, or at 7? This helps determine the nature of the acid and base involved.
Consider the shape of the curve before and after the equivalence point. A sharp rise or fall indicates a strong acid or base, while a more gradual curve suggests a weak acid or base.
Match the observed features to the possible titration types (e.g., strong acid with strong base, weak acid with strong base, etc.).
Try solving on your own before revealing the answer!
Final Answer: A weak acid is titrated with a strong base
The initial pH is below 7, and the equivalence point is above 7, which is characteristic of titrating a weak acid with a strong base.
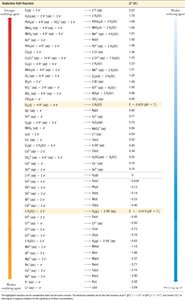
Q45. Using the table above, determine if each metal can be dissolved in HBr
Background
Topic: Electrochemistry – Activity Series and Reduction Potentials
This question tests your ability to use a table of standard reduction potentials to predict whether a metal will react with an acid (HBr) to produce hydrogen gas.
Key Terms and Concepts:
Standard Reduction Potential (E°): A measure of the tendency of a chemical species to be reduced, measured in volts.
Activity Series: Metals with lower (more negative) reduction potentials than H+ will dissolve in acid, producing H2 gas.
Relevant Reaction:
Step-by-Step Guidance
Find the standard reduction potential for each metal in the table.
Compare each metal's reduction potential to that of H+ ( V).
If the metal's reduction potential is less than 0.00 V, it will dissolve in HBr (i.e., it is a stronger reducing agent than H+).
Decide for each metal (Al, Ba, Co, Cu, Zn) whether it will dissolve in HBr based on this comparison.
Try solving on your own before revealing the answer!
Final Answer:
Aluminum: Yes
Barium: Yes
Cobalt: Yes
Copper: No
Zinc: Yes
All metals with a standard reduction potential less than 0.00 V will dissolve in HBr, except copper, which has a higher reduction potential.
