 Back
BackCHM1045 Exam 2 Study Guide: Chemical Reactions, Solutions, and Gases
Study Guide - Smart Notes

Stoichiometry and Chemical Reactions
Stoichiometry and Mole Ratios
Stoichiometry is the quantitative relationship between reactants and products in a chemical reaction. It allows chemists to predict the amounts of substances consumed and produced.
Mole ratios are derived from the coefficients in a balanced chemical equation and are used to convert between grams, moles, and particles of reactants and products.
Typical conversion sequence: grams A → moles A → moles B → grams B.
Example: In the reaction , the mole ratio between and is 1:1.
Limiting Reactant, Theoretical Yield, and Percent Yield
The limiting reactant is the substance that is completely consumed first, thus determining the maximum amount of product formed.
Theoretical yield is the maximum amount of product that can be formed from the limiting reactant.
Percent yield is calculated as:
Example: If the theoretical yield is 10 g and the actual yield is 8 g, percent yield is .
Solutions and Aqueous Chemistry
Solute vs. Solvent and Aqueous Solutions
A solution is a homogeneous mixture of two or more substances. The solute is the substance dissolved, and the solvent is the substance doing the dissolving.
Aqueous solution: A solution where water is the solvent.
Example: Salt water is an aqueous solution with NaCl as the solute and water as the solvent.
Molarity and Solution Calculations
Molarity () is a measure of concentration, defined as moles of solute per liter of solution.
Used as a conversion factor in stoichiometry and dilution calculations.
Dilution equation:
Example: To dilute 1.0 L of 2.0 M solution to 0.5 M, use L.
Electrolytes and Solubility Rules
Electrolytes are substances that dissociate into ions in solution.
Strong electrolytes: Completely dissociate (e.g., NaCl, strong acids/bases).
Weak electrolytes: Partially dissociate (e.g., weak acids/bases).
Nonelectrolytes: Do not dissociate (e.g., sugar).
Solubility rules help predict whether a compound will dissolve in water.
Precipitation Reactions and Ionic Equations
Precipitation reactions occur when two solutions are mixed and an insoluble product (precipitate) forms.
Molecular equation: Shows all compounds as intact molecules.
Complete ionic equation: Shows all strong electrolytes as ions.
Net ionic equation: Shows only the ions and molecules directly involved in the reaction.
Example: Mixing and forms precipitate.
Acid-Base Reactions and Titrations
Acid-base reactions involve the transfer of a proton () from an acid to a base.
Neutralization: Acid reacts with base to form water and a salt.
Titration: A technique to determine the concentration of an acid or base using a solution of known concentration.
Example:
Gas Evolution and Redox Reactions
Gas evolution reactions produce a gas as a product. Redox reactions involve the transfer of electrons.
Oxidation: Loss of electrons.
Reduction: Gain of electrons.
Assign oxidation numbers using rules (see below).
Identify oxidizing and reducing agents.
Example: Combustion of methane:
Properties and Laws of Gases
Gas Properties and Laws
Gases have unique properties and obey several fundamental laws.
Boyle's Law: (pressure and volume inversely related)
Charles's Law: (volume and temperature directly related)
Avogadro's Law: (volume and moles directly related)
Ideal Gas Law:
Gas constant:
STP: Standard Temperature and Pressure (0°C, 1 atm); 1 mol gas = 22.4 L
Gas Density and Molar Mass
The density of a gas at STP can be calculated using its molar mass and molar volume.
Example: at STP:
Mixtures of Gases and Partial Pressures
In a mixture, each gas exerts a partial pressure proportional to its mole fraction.
Dalton's Law:
Mole fraction:
Partial pressure:
Kinetic Molecular Theory
This theory explains the behavior of gases based on the motion of their particles.
Gas particles are in constant, random motion.
Collisions are elastic; energy is conserved.
Pressure results from collisions with container walls.
Reference Tables and Rules
Solubility Rules
Solubility rules help predict whether a compound will dissolve in water.
Group IA salts, , , are soluble.
, , salts are soluble except with , , .
salts are soluble except with , , , .
, , , salts are insoluble except with Group IA, , , .
Rules for Assigning Oxidation Numbers
Oxidation numbers are assigned to atoms in compounds to track electron transfer in redox reactions.
Sum of oxidation numbers equals the charge of the molecule or ion.
Free elements: 0
Group 1A, 2A, Al: +1, +2, +3
H: +1; F: -1
O: -2
Group 7A: -1; Group 6A: -2; Group 5A: -3
When rules conflict, use the lower number and ignore the conflicting rule.
Periodic Table of the Elements
The periodic table organizes elements by increasing atomic number and groups elements with similar properties together.
Groups (columns) indicate similar chemical properties.
Periods (rows) indicate increasing atomic number.
Metals, nonmetals, and metalloids are classified based on their properties.
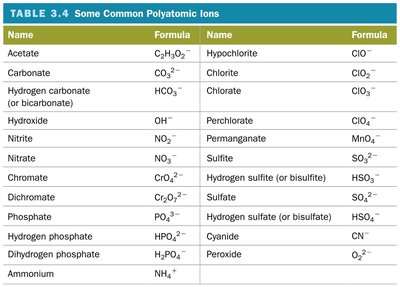
Common Polyatomic Ions
Polyatomic ions are ions composed of two or more atoms covalently bonded, carrying a net charge. They are frequently encountered in chemical reactions and formulas.
Name | Formula | Name | Formula |
|---|---|---|---|
Acetate | C2H3O2- | Hypochlorite | ClO- |
Carbonate | CO32- | Chlorite | ClO2- |
Hydrogen carbonate (bicarbonate) | HCO3- | Chlorate | ClO3- |
Hydroxide | OH- | Perchlorate | ClO4- |
Nitrite | NO2- | Permanaganate | MnO4- |
Nitrate | NO3- | Sulfite | SO32- |
Chromate | CrO42- | Hydrogen sulfite (bisulfite) | HSO3- |
Dichromate | Cr2O72- | Sulfate | SO42- |
Phosphate | PO43- | Hydrogen sulfate (bisulfate) | HSO4- |
Hydrogen phosphate | HPO42- | Cyanide | CN- |
Dihydrogen phosphate | H2PO4- | Peroxide | O22- |
Ammonium | NH4+ |
