 Back
BackColligative Properties and Raoult's Law: Vapor Pressure of Solutions
Study Guide - Smart Notes

Colligative Properties
Definition and Importance
Colligative properties are properties of mixtures that depend on the total number of dissolved particles, but not necessarily on the identity of the solute. These properties are crucial in understanding how solutions behave, especially in relation to their physical characteristics such as vapor pressure, boiling point, freezing point, and osmotic pressure.
Key Point: Colligative properties are determined by the concentration of solute particles, not their chemical nature.
Examples: Lowering of vapor pressure, boiling point elevation, freezing point depression, and osmotic pressure.
Raoult's Law
Vapor Pressure of Solutions
Raoult's Law describes how the vapor pressure of a solution is related to the mole fraction of the solvent and the vapor pressure of the pure solvent. It is especially applicable to ideal solutions, where the interactions between solute and solvent are similar to those in the pure substances.
For a non-volatile solute: The solute does not evaporate, so only the solvent contributes to the vapor pressure.
Raoult's Law Equation:
: Vapor pressure of the solution
: Mole fraction of the solvent
: Vapor pressure of the pure solvent
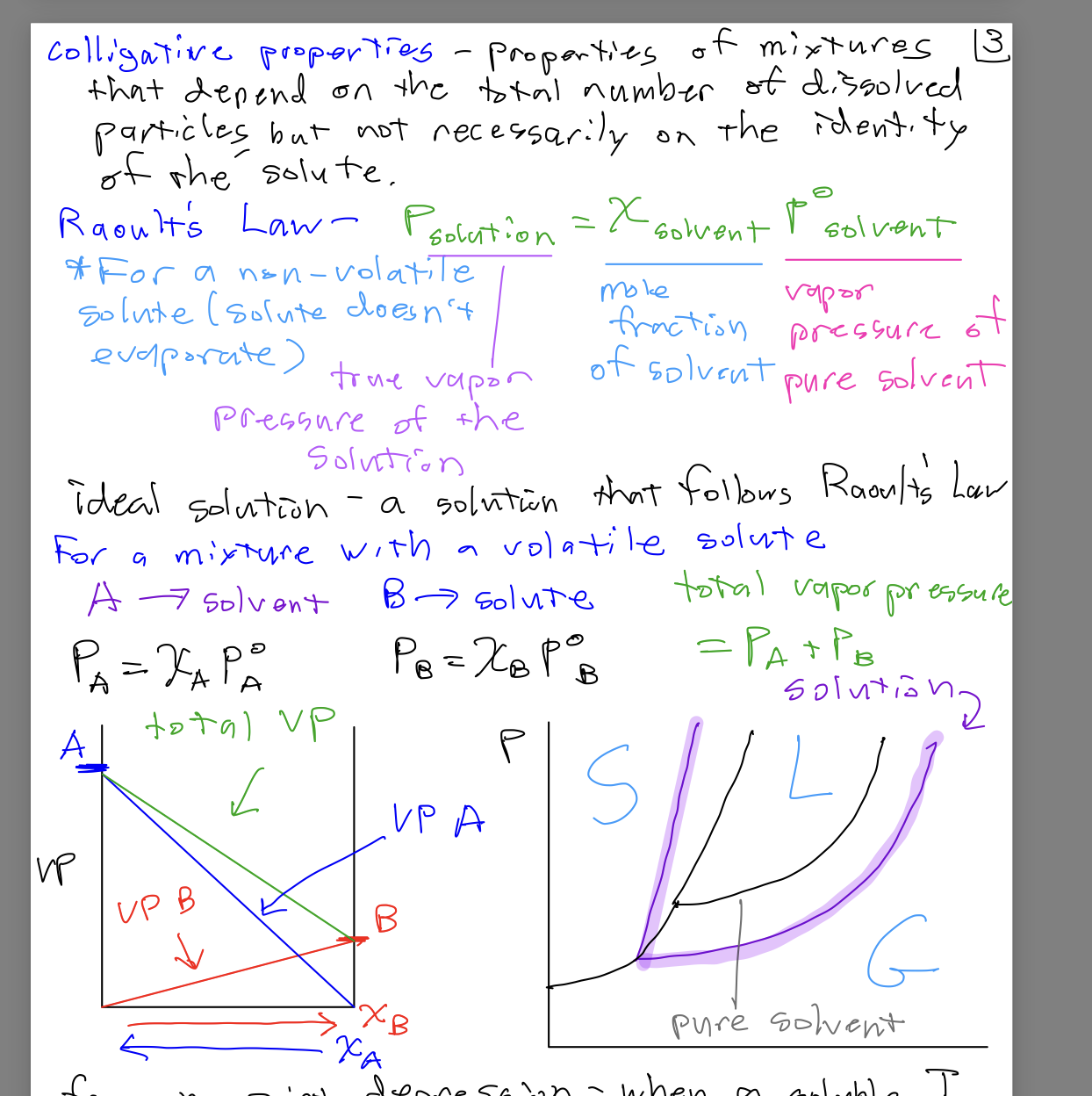
Ideal Solutions and Volatile Solutes
An ideal solution is one that follows Raoult's Law exactly. For mixtures with volatile solutes, both the solvent and solute contribute to the total vapor pressure.
For a mixture with a volatile solute:
: Partial vapor pressure of component A (solvent)
: Partial vapor pressure of component B (solute)
and : Mole fractions of A and B
and : Vapor pressures of pure A and B
Total vapor pressure: Sum of partial pressures from each component
Graphical Representation
Vapor pressure diagrams illustrate how the vapor pressure of each component and the total vapor pressure change with composition. In ideal solutions, the total vapor pressure is a linear combination of the partial pressures, weighted by mole fraction.
VP vs. Mole Fraction: Shows how vapor pressure varies as the composition of the solution changes.
Phase Diagrams: Indicate the regions of solid (S), liquid (L), and gas (G) phases, and how the presence of solute affects these boundaries.
Summary Table: Raoult's Law for Solutions
Type of Solution | Raoult's Law Equation | Key Feature |
|---|---|---|
Non-volatile solute | Only solvent contributes to vapor pressure | |
Volatile solute | Both solvent and solute contribute | |
Ideal solution | Follows Raoult's Law exactly | Linear relationship between vapor pressure and composition |
Additional info: Colligative properties are fundamental in understanding solution behavior and are widely applied in fields such as chemical engineering, environmental science, and biochemistry.
