 Back
BackL16: Colligative Properties and Reaction Rates: Study Notes for General Chemistry
Study Guide - Smart Notes

Colligative Properties of Solutions
Introduction to Colligative Properties
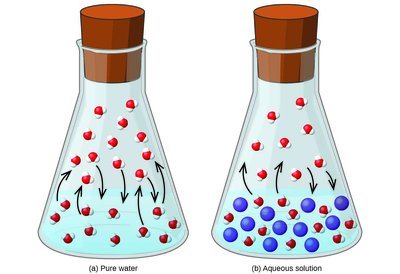
Colligative properties are physical properties of solutions that depend on the concentration of solute particles, not their identity. These include vapor pressure lowering, boiling point elevation, freezing point depression, and osmotic pressure. Understanding these properties is essential for predicting how solutions behave compared to pure solvents.
Vapor Pressure Lowering and Raoult’s Law
When a non-volatile solute is dissolved in a solvent, the vapor pressure of the solution is lower than that of the pure solvent. Raoult’s Law quantifies this effect:
Raoult’s Law (for non-volatile solutes): , where is the vapor pressure of the solution, is the mole fraction of the solvent, and is the vapor pressure of the pure solvent.
For volatile solutes: and ,
Ideal solutions: Occur when intermolecular forces (IMFs) between all components are similar.
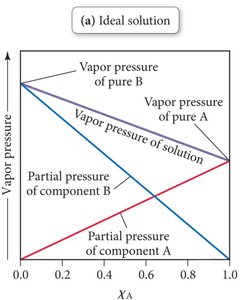
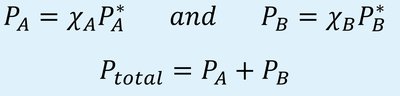
Non-Ideal Solutions: Intermolecular Forces
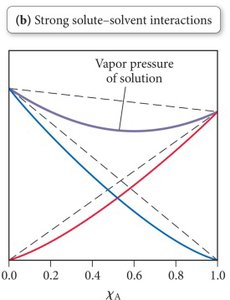
Non-ideal solutions arise when solute-solvent interactions differ from solvent-solvent or solute-solute interactions. This affects the vapor pressure:
Strong solute-solvent interactions: Lower vapor pressure than predicted by Raoult’s Law.
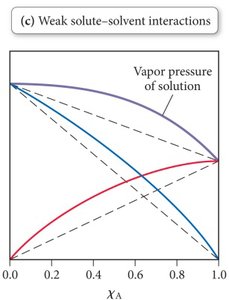
Weak solute-solvent interactions: Higher vapor pressure than predicted by Raoult’s Law.
Examples: Acetone and chloroform (strong interaction, low vapor pressure); benzene and methanol (weak interaction, high vapor pressure).
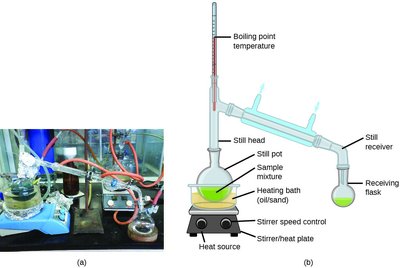
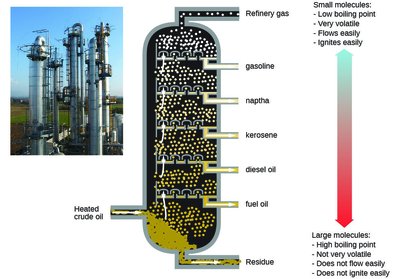
Distillation and Fractional Distillation
Distillation is a technique used to separate mixtures based on differences in boiling points. Fractional distillation allows for the separation of multiple components by repeated vaporization and condensation.
Principle: The component with the lowest boiling point vaporizes first.
Applications: Purification of liquids, separation of crude oil into fractions.
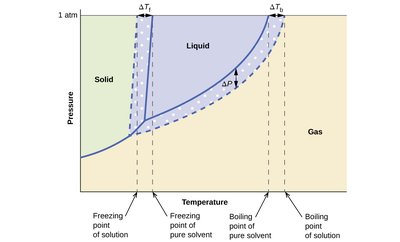
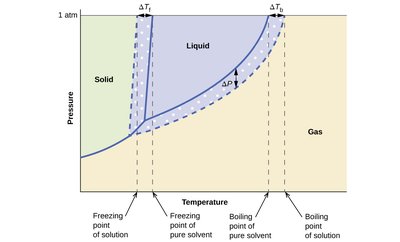
Boiling Point Elevation and Freezing Point Depression
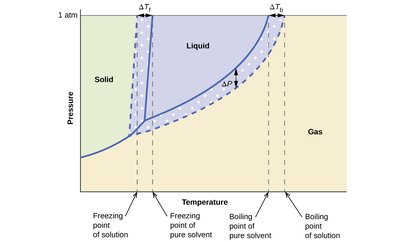
Adding solute to a solvent changes its phase change temperatures:
Boiling Point Elevation: The boiling point increases as solute is added. Formula:
Freezing Point Depression: The freezing point decreases as solute is added. Formula:
Molality (m): Moles of solute per kilogram of solvent.
Phase diagram: Shows shifts in boiling and freezing points due to solute addition.
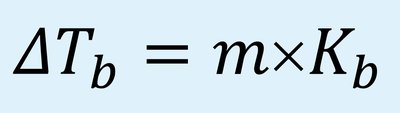
Osmosis and Osmotic Pressure
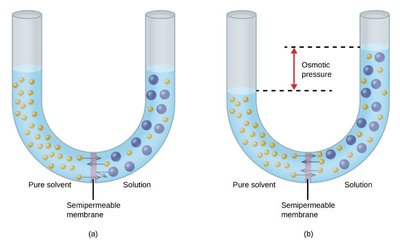
Osmosis is the movement of solvent molecules through a semipermeable membrane from a region of lower solute concentration to higher concentration. Osmotic pressure is the pressure required to stop this flow.
Osmotic Pressure Formula:
Van’t Hoff Factor (i): Accounts for the number of particles formed by dissociation of solute.
Applications: Biological membranes, water purification.
Reaction Rates
Introduction to Reaction Rates
The rate of a chemical reaction measures how quickly reactants are converted to products. It is often expressed as the change in concentration of a reactant or product per unit time.
Rate Expression:
Units: Typically molarity per second (M/s).
Relative Rates and Stoichiometry
Reaction rates can be defined for any reactant or product, but must be adjusted for stoichiometric coefficients:
Positive for products, negative for reactants.
Divide by stoichiometric coefficient: For ,
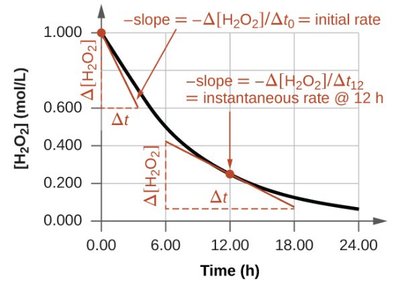
Average and Instantaneous Rates
Average rate is calculated over a time interval, while instantaneous rate is the rate at a specific moment, often determined by the slope of a concentration vs. time graph.
Average Rate: over a time interval.
Instantaneous Rate: Slope of the tangent to the curve at a specific time.
Key Terms and Concepts
Colligative Properties: Properties that depend on the number of solute particles.
Raoult’s Law: Describes vapor pressure lowering in solutions.
Van’t Hoff Factor: Number of particles formed by solute dissociation.
Osmosis: Movement of solvent across a membrane.
Reaction Rate: Change in concentration of reactants/products per unit time.
