 Back
BackChapter 14 lecture 3
Study Guide - Smart Notes

Colligative Properties of Solutions
Introduction to Colligative Properties
Colligative properties are physical properties of solutions that depend on the number of solute particles present, not their chemical identity. These properties are influenced by whether the solute is an electrolyte (dissociates into ions) or a nonelectrolyte (does not dissociate).
Key Colligative Properties: Vapor pressure lowering, boiling point elevation, freezing point depression, osmotic pressure, and the van’t Hoff factor.
Electrolyte vs. Nonelectrolyte: Electrolytes produce more particles in solution, affecting colligative properties more strongly than nonelectrolytes.
Vapor Pressure Lowering
Raoult’s Law and the Effect of Nonvolatile Solutes
The addition of a nonvolatile solute to a solvent lowers the vapor pressure of the solution compared to the pure solvent. This occurs because fewer solvent molecules are present at the surface to escape into the vapor phase.
Raoult’s Law:
Vapor Pressure Lowering:
Entropy Explanation: The solution is more disordered than the pure solvent, so the difference in entropy between solution and vapor is less, reducing the tendency for solvent molecules to vaporize.
Example: Calculating vapor pressure lowering for solutions of glycerin and glucose in water.
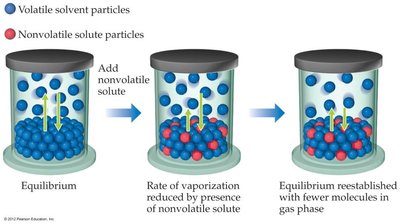
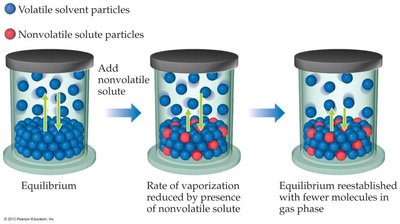
Vapor Pressure of Solutions with Volatile Solutes
In solutions with two volatile components, both contribute to the vapor pressure. Raoult’s Law applies to each component, and the total vapor pressure is the sum of the partial pressures.
Raoult’s Law for Each Component: ,
Total Pressure:
Deviations from Raoult’s Law: Strong solute-solvent interactions cause negative deviations (lower vapor pressure), while weak interactions cause positive deviations (higher vapor pressure).
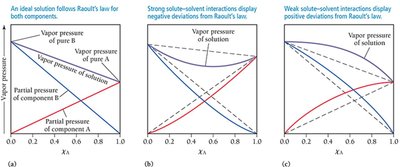
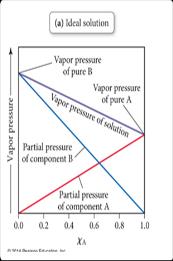
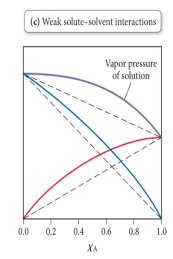
Boiling Point Elevation
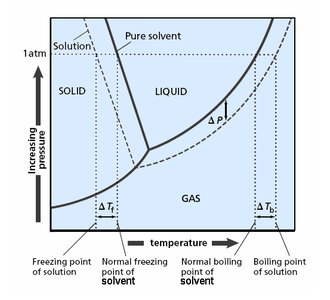
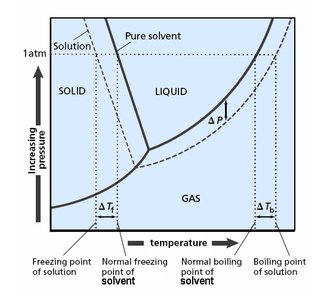
Phase Diagram and Boiling Point Changes
The addition of a nonvolatile solute lowers the vapor pressure, requiring a higher temperature to reach boiling. Thus, the boiling point of the solution is elevated compared to the pure solvent.
Boiling Point Elevation Equation:
Molality (m): Moles of solute per kilogram of solvent.
Boiling Point Elevation Constant (k_b): Depends only on the solvent, units are °C/m.
Example: Calculating boiling point elevation for glucose in water.
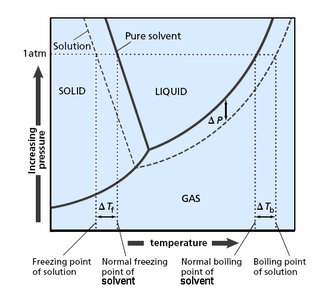
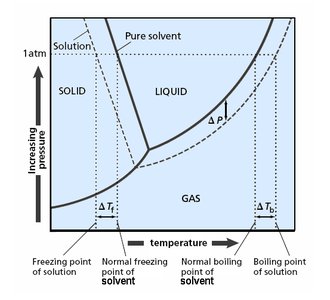
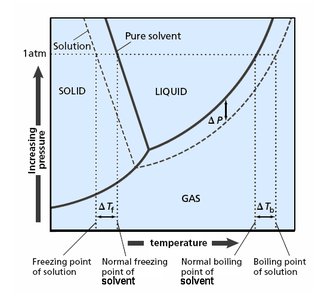
Freezing Point Depression
Phase Diagram and Freezing Point Changes
The addition of a nonvolatile solute lowers the freezing point of the solution compared to the pure solvent. This is because the solute disrupts the formation of the solid phase.
Freezing Point Depression Equation:
Molal Freezing Point Depression Constant (k_f): Depends only on the solvent, units are °C/m.
Example: Calculating freezing point depression for glucose in water.
Application: Cryoscopy is used to determine molar mass by measuring freezing point depression.
Osmotic Pressure
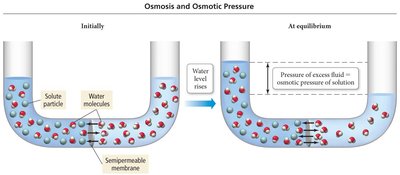
Osmosis and Osmotic Pressure
Osmosis is the movement of solvent through a semipermeable membrane to equalize solute concentration. Osmotic pressure is the pressure required to stop this flow.
Osmotic Pressure Equation:
Variables: = osmotic pressure (atm), = molarity, = gas constant (0.0821 L·atm/K·mol), = temperature (K).
Applications: Reverse osmosis for desalination, biological cell environments (hypertonic, hypotonic, isotonic).
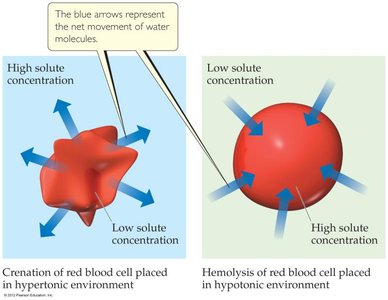
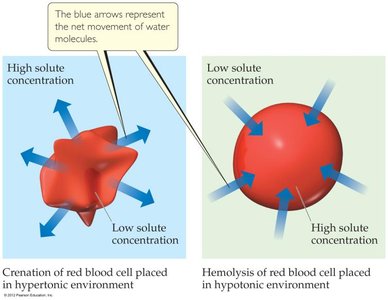
van’t Hoff Factor (i)
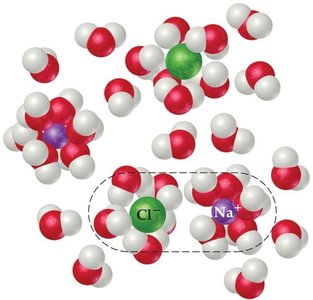
Electrolyte Solutions and Colligative Properties
The van’t Hoff factor accounts for the number of particles produced by dissociation of electrolytes in solution. Colligative property equations are modified by multiplying by i.
van’t Hoff Factor Equation:
Modified Colligative Property Equations:
Example: Freezing point depression for CuCl2 in water, with i = 3.
Experimental vs. Theoretical i: Ion pairing at higher concentrations reduces the effective number of particles, causing deviations from the theoretical value.
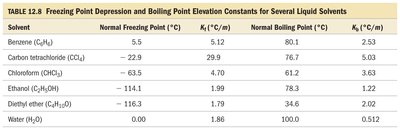
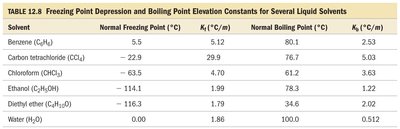
Summary Table: Freezing Point Depression and Boiling Point Elevation Constants
The following table summarizes the freezing point depression and boiling point elevation constants for several common solvents:
Solvent | Normal Freezing Point (°C) | kf (°C/m) | Normal Boiling Point (°C) | kb (°C/m) |
|---|---|---|---|---|
Benzene (C6H6) | 5.5 | 5.12 | 80.1 | 2.53 |
Carbon tetrachloride (CCl4) | -22.9 | 29.9 | 76.7 | 5.03 |
Chloroform (CHCl3) | -63.5 | 4.70 | 61.2 | 3.63 |
Ethanol (C2H5OH) | -114.1 | 1.99 | 78.3 | 1.22 |
Diethyl ether (C4H10O) | -116.3 | 1.79 | 34.6 | 2.02 |
Water (H2O) | 0.00 | 1.86 | 100.0 | 0.512 |
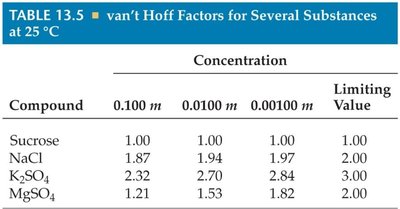
Summary Table: van’t Hoff Factors for Several Substances
The following table shows van’t Hoff factors for several substances at different concentrations:
Compound | 0.100 m | 0.0100 m | 0.00100 m | Limiting Value |
|---|---|---|---|---|
Sucrose | 1.00 | 1.00 | 1.00 | 1.00 |
NaCl | 1.87 | 1.94 | 1.97 | 2.00 |
K2SO4 | 2.32 | 2.70 | 2.84 | 3.00 |
MgSO4 | 1.21 | 1.53 | 1.82 | 2.00 |
Additional info: This study guide expands on lecture notes with full academic explanations, equations, and relevant examples for each colligative property. All included images and tables directly reinforce the concepts discussed.
