 Back
BackComprehensive Study Notes on Gases – Properties, Laws, and Applications
Study Guide - Smart Notes

Properties of Gases and Gas Pressure
Physical Properties of Gases
Gases are a state of matter characterized by their ability to expand and fill the shape of their containers, mix evenly and completely with other gases, and be compressible. Their physical behavior is described by four main variables: pressure (P), volume (V), temperature (T), and amount (n, in moles).
Expansion: Gases expand to fill their containers.
Mixing: Gases diffuse and mix in all proportions.
Visibility: Some gases can be seen if colored.

The Concept of Pressure
Pressure is defined as the force exerted per unit area. In gases, pressure results from collisions of gas particles with the walls of their container.
Mathematical Definition: , where is force and is area.
SI Unit: Pascal (Pa), where .
Liquid Pressure: , where is density, is height, and is acceleration due to gravity.

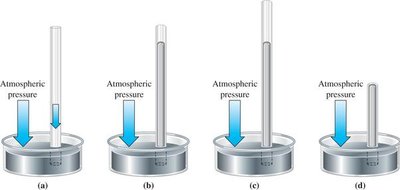
Barometric Pressure
Barometric pressure is the pressure exerted by the atmosphere and is commonly measured using a barometer. At sea level and 0°C, the standard atmospheric pressure is defined as 760 mmHg (or 1 atm).
Comparing Liquid Pressures
The height of a liquid column that exerts the same pressure as a column of mercury can be calculated using the relationship between density and height. This is important for comparing different manometric fluids.

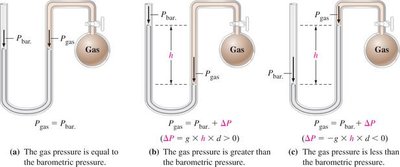
Manometers
Manometers are devices used to measure the pressure of a gas in a container. They compare the gas pressure to atmospheric pressure using a column of liquid (often mercury).

Pressure Units
Several units are used to express gas pressure:
Atmosphere (atm): Standard atmospheric pressure.
Millimeter of mercury (mmHg) or Torr: 1 atm = 760 mmHg = 760 Torr.
Pascals (Pa): 1 atm = 101,325 Pa.
Bar: 1 bar = 100,000 Pa.
Using SI Units of Pressure
Pressure can be calculated in SI units and converted to other units as needed. The force exerted by a mass and the area over which it acts are used to determine pressure in pascals, which can then be converted to torr or atm.

The Simple Gas Laws
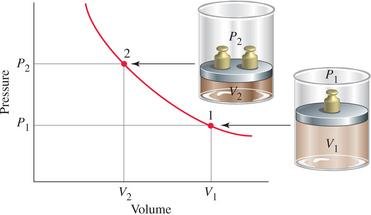
Boyle’s Law
Boyle’s Law states that for a fixed amount of gas at constant temperature, the volume of the gas is inversely proportional to its pressure.
Mathematical Form: or
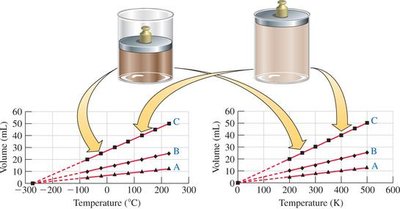
Charles’s Law
Charles’s Law states that the volume of a fixed amount of gas at constant pressure is directly proportional to its absolute temperature (in Kelvin).
Mathematical Form: or
Avogadro’s Law
Avogadro’s Law states that at constant temperature and pressure, the volume of a gas is directly proportional to the number of moles of gas present.
Mathematical Form: or
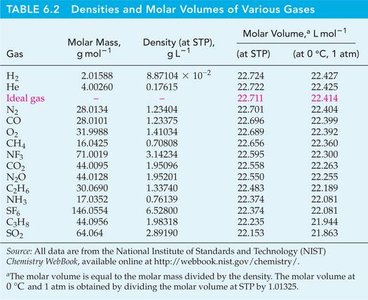
Standard Temperature and Pressure (STP)
STP is defined as 0°C (273.15 K) and 1 bar (100 kPa). At STP, 1 mole of an ideal gas occupies 22.711 L.
Combining the Gas Laws and the Ideal Gas Equation
General Gas Law
The combined gas law incorporates Boyle’s, Charles’s, and Avogadro’s laws:
The Ideal Gas Law is:
Where is the universal gas constant. Common values for include:

Applications of the Ideal Gas Equation
The ideal gas equation can be used to solve for any one variable if the others are known. It is also used to determine molar mass, density, and stoichiometric relationships in chemical reactions involving gases.





Gases in Chemical Reactions
Stoichiometry and Gas Laws
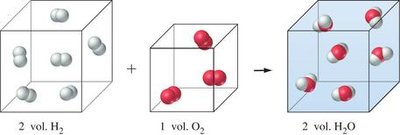
Gas laws are used to relate volumes of gases in chemical reactions. The law of combining volumes states that gases react in simple whole-number ratios by volume under the same conditions of temperature and pressure.


Mixtures of Gases
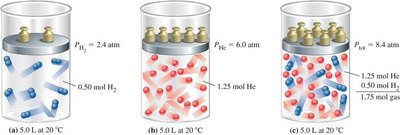
Dalton’s Law of Partial Pressures
Dalton’s Law states that the total pressure exerted by a mixture of non-reacting gases is equal to the sum of the partial pressures of each individual gas.
The partial pressure of a gas is proportional to its mole fraction in the mixture:
Mole fraction:


Kinetic-Molecular Theory of Gases
Basic Postulates
The kinetic-molecular theory explains the behavior of gases based on the motion of their particles:
Gas particles are in constant, random motion.
The volume of individual gas particles is negligible compared to the volume of the container.
Collisions between gas particles and with the container walls are perfectly elastic.
There are no intermolecular forces between gas particles.
The average kinetic energy of gas particles is proportional to the absolute temperature.
The root-mean-square speed () of gas molecules is given by:
Diffusion and Effusion
Diffusion is the mixing of gases due to molecular motion. Effusion is the process by which gas molecules escape through a tiny hole into a vacuum. Graham’s Law describes the rate of effusion:
For two gases,
Nonideal (Real) Gases
Deviations from Ideal Behavior
Real gases deviate from ideal behavior at high pressures and low temperatures due to intermolecular forces and the finite volume of gas particles. The van der Waals equation accounts for these deviations:
Where corrects for intermolecular attractions and corrects for the finite volume of molecules.
The van der Waals equation is most accurate for gases with nearly spherical molecules and small dipole moments.
