 Back
BackCounting π-Bonds in an Organic Molecule
Study Guide - Smart Notes

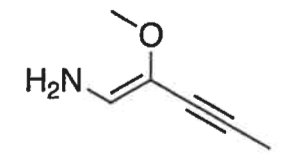
Q1. How many π-bonds are in the following molecule?
Background
Topic: Bond Types in Organic Molecules
This question tests your ability to identify and count π-bonds (pi bonds) in a molecule. π-bonds are found in double and triple bonds, and are important in understanding molecular structure and reactivity.
Key Terms and Concepts:
π-bond (pi bond): A type of covalent bond formed by sideways overlap of p orbitals. Each double bond contains one π-bond, and each triple bond contains two π-bonds.
Single bond: Contains only a sigma (σ) bond.
Double bond: Contains one sigma (σ) and one π-bond.
Triple bond: Contains one sigma (σ) and two π-bonds.
Step-by-Step Guidance
Examine the molecule and identify all double and triple bonds present.
For each double bond, count one π-bond.
For each triple bond, count two π-bonds.
Add up the total number of π-bonds from all double and triple bonds in the molecule.
Try solving on your own before revealing the answer!
Final Answer: 3 π-bonds
There is one double bond (1 π-bond) and one triple bond (2 π-bonds), for a total of 3 π-bonds.
This is a common approach for counting π-bonds in organic molecules: double bonds contribute one, triple bonds contribute two.
