 Back
BackCrystalline Solids and Modern Materials: Structure and Properties
Study Guide - Smart Notes

Crystalline Solids and Modern Materials
Classification of Solids
Solids are classified based on the arrangement of their constituent particles. The two main types are crystalline solids and amorphous solids.
Crystalline Solid: Atoms, ions, or molecules are arranged in a highly ordered, repeating pattern throughout the material.
Amorphous Solid: Particles are randomly arranged with no discernible long-range order or pattern.
Example: Olive oil hardens over a range of temperatures, indicating it is an amorphous solid.
Types of Solids
Solids can be further classified based on their smallest units and the forces holding them together:
Ionic Solids: Composed of ions held together by electrostatic attraction between cations and anions. Example: AlF3.
Molecular Solids: Made of molecules held together by intermolecular forces (e.g., hydrogen bonding, dipole-dipole, London dispersion). Example: CO2.
Covalent Network Solids: Atoms connected by covalent bonds in a continuous network. Example: Graphite.
Metallic Solids: Metal atoms held together by metallic bonds (pooling of electrons). Example: Steel, Bronze.
Amorphous Solids: No long-range order; can include plastics.
Properties Comparison:
Ionic: Hard, high melting point, brittle, conducts electricity when molten.
Molecular: Soft, low melting point, poor conductors.
Covalent Network: Hard, very high melting point, poor conductors.
Metallic: Shiny, malleable, variable melting points, good conductors.
Amorphous: Variable hardness and melting point, no distinct melting point.
Practice: Classify the following:
Steel: Metallic solid
CO2: Molecular solid
Graphite: Covalent network solid
CaCO3: Ionic solid
Bronze: Metallic alloy
Crystalline Solids: Structure and Unit Cells
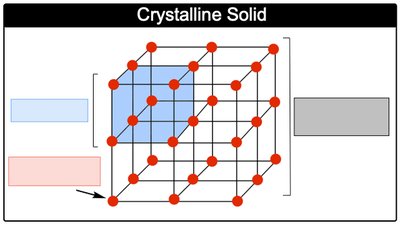
Crystal Lattice and Unit Cell
Crystalline solids are composed of a repeating three-dimensional arrangement called a crystal lattice. The unit cell is the smallest repeating structural unit of the lattice.
Unit Cell: The basic building block of the crystal lattice.
Lattice Point: A position in the lattice occupied by an atom, ion, or molecule.

Cubic Unit Cells
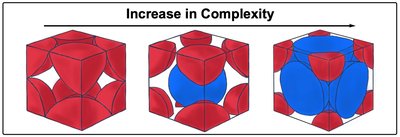
There are three main types of cubic unit cells, each with different coordination numbers and packing efficiencies:
Simple Cubic (SC): Atoms at each corner of the cube.
Body-Centered Cubic (BCC): Atoms at each corner and one atom in the center.
Face-Centered Cubic (FCC): Atoms at each corner and one atom at the center of each face.
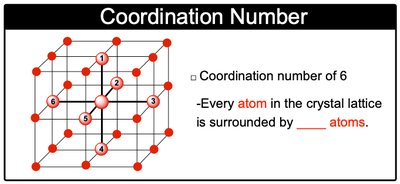
Coordination Number: The number of nearest neighbor atoms surrounding a given atom in the lattice.
Packing Efficiency: The percentage of space occupied by atoms in the unit cell.
As complexity increases, coordination number and packing efficiency increase.
Types of Cubic Unit Cells
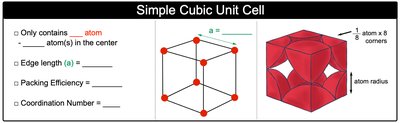
Simple Cubic Unit Cell (SC)
The simple cubic unit cell contains atoms at each corner of the cube. It is the least efficient packing arrangement.
Number of atoms per unit cell: 1 (each corner atom is shared by 8 unit cells)
Coordination number: 6
Packing efficiency: 52%
Edge length (a): (where is the atomic radius)
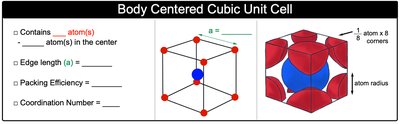
Body-Centered Cubic Unit Cell (BCC)
The body-centered cubic unit cell contains atoms at each corner and one atom in the center of the cube.
Number of atoms per unit cell: 2 (1 from corners, 1 from center)
Coordination number: 8
Packing efficiency: 68%
Edge length (a):
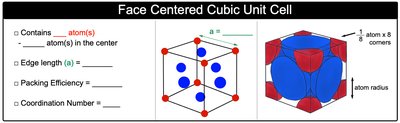
Face-Centered Cubic Unit Cell (FCC)
The face-centered cubic unit cell contains atoms at each corner and at the center of each face.
Number of atoms per unit cell: 4 (1 from corners, 3 from faces)
Coordination number: 12
Packing efficiency: 74%
Edge length (a):
Summary Table: Cubic Unit Cells
Type | Atoms per Unit Cell | Coordination Number | Packing Efficiency (%) | Edge Length Formula |
|---|---|---|---|---|
Simple Cubic (SC) | 1 | 6 | 52 | |
Body-Centered Cubic (BCC) | 2 | 8 | 68 | |
Face-Centered Cubic (FCC) | 4 | 12 | 74 |
Practice Problems and Applications
Calculate the number of atoms in one mole of a simple cubic element given its density, radius, and molar mass.
Determine the density or edge length of a unit cell given atomic radius and structure type.
Classify solids based on their properties and structure.
Additional info: The notes include practice questions and examples to reinforce understanding of unit cell calculations and solid classification. The images provided visually clarify the structure and packing of different cubic unit cells, coordination numbers, and the concept of increasing complexity in crystal lattices.
