 Back
BackCrystalline Solids: Structure, Classification, and Properties
Study Guide - Smart Notes

Crystalline Solids and X-Ray Diffraction
X-Ray Diffraction and Crystallography
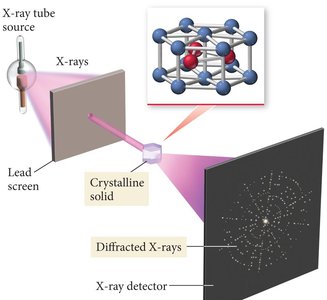
Crystalline solids possess a highly ordered arrangement of atoms, molecules, or ions. The structure of these solids can be determined using X-ray diffraction, a technique that reveals the atomic arrangement by analyzing the pattern produced when X-rays are scattered by the crystal.
X-ray diffraction is used to determine the atomic structure of crystalline solids.
When X-rays pass through a crystal, they are diffracted, producing a pattern that can be analyzed to reveal the positions of atoms.
By rotating the crystal and collecting multiple diffraction patterns, the full atomic structure can be reconstructed.
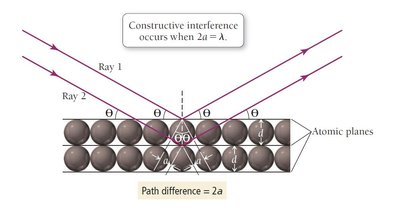
Bragg's Law
Constructive interference of X-rays occurs at specific angles, which are related to the separation between atomic planes, the wavelength of the X-rays, and the order of diffraction. This relationship is described by Bragg's law:
Bragg's law:
n: Order of diffraction (usually 1 unless specified)
d: Distance between atomic planes
\lambda: Wavelength of X-rays
\theta: Angle of incidence
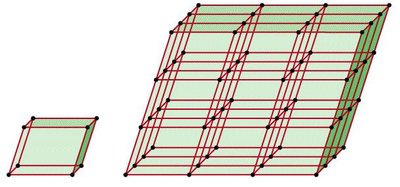
Crystal Lattice and Unit Cell
Crystal Lattice and Unit Cell
The crystal lattice is a three-dimensional arrangement of points representing the positions of atoms, molecules, or ions in a crystalline solid. The unit cell is the smallest repeating structural unit that defines the entire lattice.
Lattice points are occupied by atoms, molecules, or ions.
The unit cell is the basic repeating unit of the crystal lattice.
Crystalline solids exhibit long-range order, meaning their structure repeats predictably throughout the material.
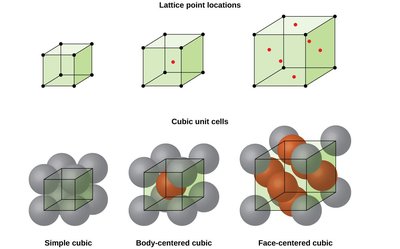
Types of Unit Cells
There are several common types of unit cells in crystalline solids, each with distinct arrangements and properties. The most important cubic unit cells are simple cubic, body-centered cubic (BCC), and face-centered cubic (FCC).
Simple cubic: Atoms at each corner of the cube.
Body-centered cubic (BCC): Atoms at each corner and one at the center of the cube.
Face-centered cubic (FCC): Atoms at each corner and at the center of each face.
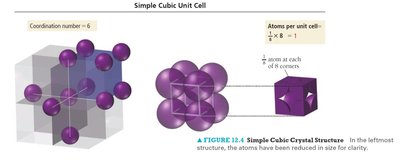
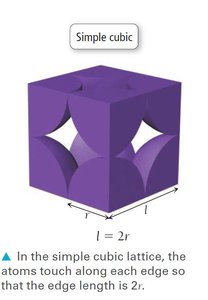
Simple Cubic Unit Cell
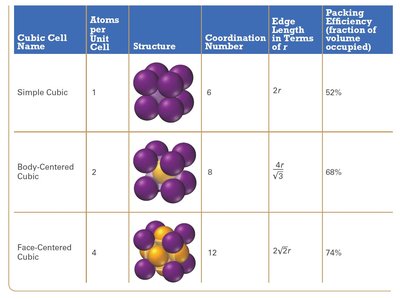
The simple cubic unit cell is the most basic type, with atoms located at each corner. It has a low packing efficiency and a coordination number of 6.
Coordination number: 6 (each atom touches 6 neighbors)
Atoms per unit cell: 1 (each corner atom is shared by 8 unit cells)
Packing efficiency: 52% (fraction of volume occupied by atoms)
Body-Centered Cubic (BCC) Unit Cell
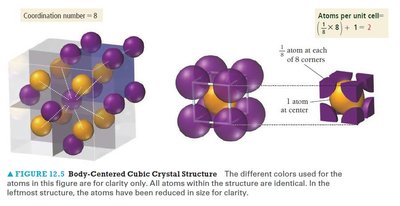
The BCC unit cell has atoms at each corner and one at the center. It has a higher packing efficiency and coordination number than the simple cubic cell.
Coordination number: 8
Atoms per unit cell: 2
Packing efficiency: 68%
Face-Centered Cubic (FCC) Unit Cell
The FCC unit cell has atoms at each corner and at the center of each face. It has the highest packing efficiency and coordination number among cubic cells.
Coordination number: 12
Atoms per unit cell: 4
Packing efficiency: 74%
Close Packing of Spheres
Hexagonal Close Packing (HCP)
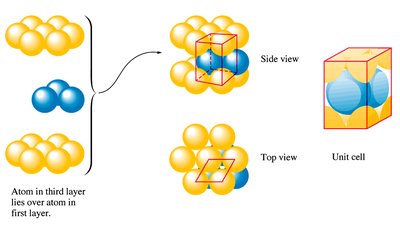
Close packing refers to the arrangement of spheres (atoms) to maximize packing efficiency. The hexagonal close packed (hcp) structure follows an ABAB pattern, resulting in a coordination number of 12.
HCP: ABAB pattern of atomic planes
Coordination number: 12
Common in metals, minimizing wasted space
Cubic Close Packing (CCP)
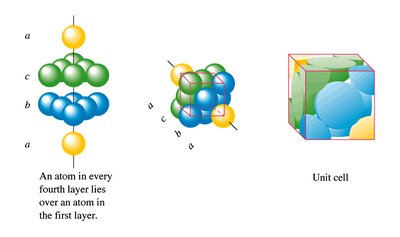
The cubic close packed (ccp) structure follows an ABCABC pattern and is equivalent to the FCC unit cell. It also achieves a coordination number of 12 and 74% packing efficiency.
CCP: ABCABC pattern
Equivalent to FCC unit cell
High packing efficiency
Classification of Solids
Amorphous vs. Crystalline Solids
Solids can be classified based on their degree of order. Amorphous solids lack long-range order, while crystalline solids have a highly ordered arrangement.
Amorphous solids: No well-defined long-range arrangement; short-range order only.
Crystalline solids: Atoms arranged in a highly ordered, repeating pattern.



Types of Ordered Solids
Ordered solids are further classified based on the nature of their lattice points and the forces holding them together.
Molecular solids: Lattice points occupied by molecules, held together by weak intermolecular forces. Soft, low melting points, poor conductors.
Ionic solids: Lattice points occupied by ions, held together by electrostatic attraction. Hard, brittle, high melting points, poor conductors.
Atomic solids: Lattice points occupied by atoms. Can be nonbonding, metallic, or network covalent.



Molecular Solids
Molecular solids are composed of molecules at lattice points, held together by intermolecular forces. They are typically soft, have low melting points, and are poor conductors.
Examples: solid CO2 (dry ice), ice
Can form different crystal structures called polymorphs

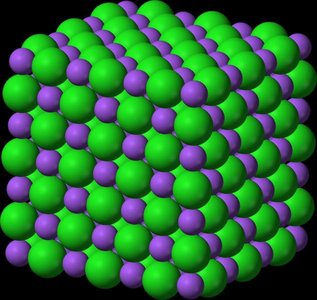
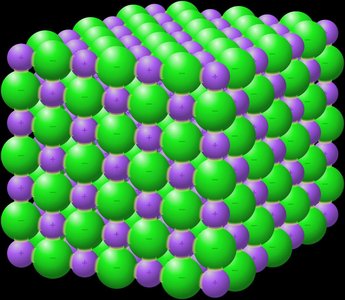
Ionic Solids
Ionic solids consist of cations and anions at lattice points, held together by electrostatic forces. They are hard, brittle, have high melting points, and are poor conductors.
Example: NaCl (table salt)
Complex unit cells possible
Atomic Solids
Atomic solids have atoms at lattice points and are classified as nonbonding, metallic, or network covalent based on the bonding and structure.
Nonbonding: Held by dispersion forces, close-packed, formed by noble gases.
Metallic: Metal ions in a sea of mobile electrons, variable hardness and melting points, good conductors.
Network covalent: Held by covalent bonds, hard, very high melting points, poor conductors (except graphite).

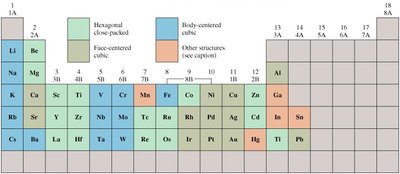
Metallic Atomic Solids
Metallic solids are formed by metal atoms, with a structure characterized by a "sea" of mobile electrons. They are typically good conductors and adopt close-packed structures.
Formed by most group 1, 2, 3, and transition elements
Variable hardness and melting points

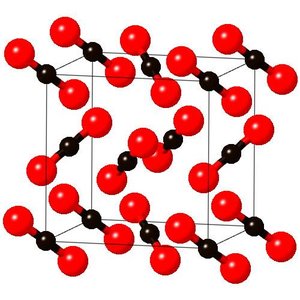
Covalent Network Solids
Covalent network solids are held together by covalent bonds, resulting in very hard materials with high melting points. They are usually formed by nonmetals with valence greater than 2 or their oxides.
Examples: Quartz (SiO2), Silicon (Si), Diamond (C)
Exception: Graphite conducts electricity



Allotropes of Carbon
Some elements, such as carbon, exist in different structural forms called allotropes. Diamond and graphite are two common allotropes of carbon, each with distinct properties.
Diamond: Each carbon atom forms four covalent bonds, resulting in a very hard structure with a high melting point (~4000 °C). Does not conduct electricity.
Graphite: Each carbon atom forms three covalent bonds, with the fourth electron delocalized across the sheet. Excellent electrical conductor, high melting point, very soft.
Additional info: Allotropes are important in materials science and chemistry due to their varied physical and chemical properties.
