 Back
BackDistinguishing Substances: Chemical Thinking and the Particulate Model
Study Guide - Smart Notes

Unit 1: How Do We Distinguish Substances?
Introduction
The ability to distinguish between different substances is fundamental in chemistry and has broad applications in fields such as food control, pollution detection, health monitoring, crime investigation, drug development, and resource management. Failure to accurately identify substances can have significant consequences, including health risks, environmental damage, and legal issues.
Food Control: Ensures safety and quality of consumables.
Pollution Detection: Identifies harmful substances in the environment.
Health Monitoring: Detects biomarkers and contaminants in biological samples.
Crime Investigation: Analyzes evidence for forensic purposes.
Drug Development: Characterizes compounds for pharmaceutical use.
Resource Exploration: Identifies valuable materials in mining and management.





Module 1: Searching for Differences
Distinguishing Characteristics
To identify and separate substances, chemists rely on differentiating characteristics—properties that are unique to each substance. These include physical and chemical properties such as boiling point, density, solubility, and mass.
Detection: Recognizing the presence of a substance.
Separation: Isolating components based on unique properties.
Identification: Determining the identity of a substance.
Quantification: Measuring the amount of a substance.
Good differentiating characteristics should be measurable, reproducible, and specific to the substance.
Classification of Matter
All matter is classified as either pure substances or mixtures:
Pure Substances: Constant composition; can be elements or compounds.
Mixtures: Composed of two or more substances in varying amounts.
Heterogeneous Mixtures: Composition varies from point to point.
Homogeneous Mixtures: Uniform composition throughout.
Phase Behavior and Separation Techniques
Phase properties, such as boiling and condensation points, are often used to separate components of mixtures. Understanding phase transitions is essential for successful separation.
Filtration: Separates solids from liquids based on particle size.
Distillation: Separates liquids based on differences in boiling points.
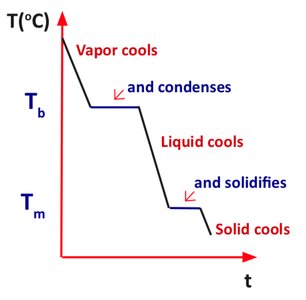
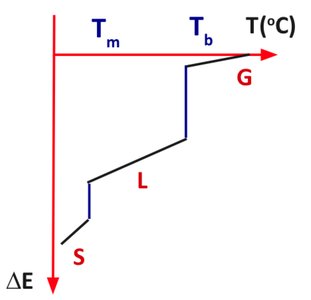
Phase Transitions
Phase transitions involve changes in physical state (solid, liquid, gas) and are accompanied by energy transfer between the system and surroundings.
Energy Added (+): Endothermic processes (melting, vaporization).
Energy Released (-): Exothermic processes (condensation, freezing).
Energy is measured in joules (J) and calories (cal):
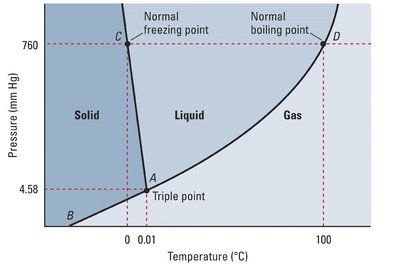
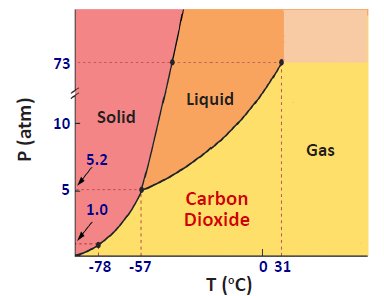
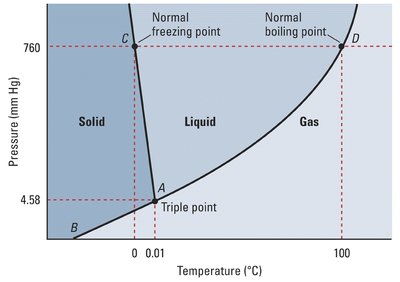
Phase Diagrams
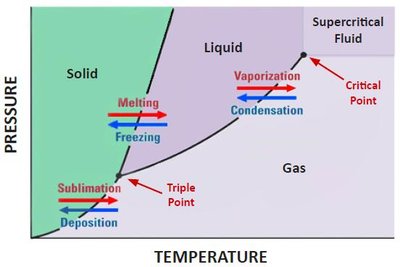
Phase diagrams illustrate the stability of different phases at various temperatures and pressures. Key points include:
Triple Point: All three phases coexist.
Critical Point: Liquid and gas phases become indistinguishable (supercritical fluid).
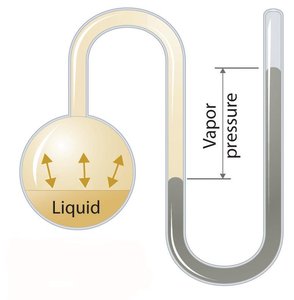
Boiling and Vapor Pressure
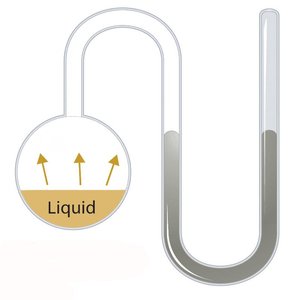
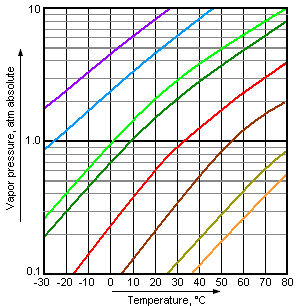
Boiling occurs when a liquid's vapor pressure equals atmospheric pressure. Vapor pressure is the pressure exerted by a vapor in equilibrium with its liquid or solid phase.
Volatility: More volatile liquids have higher vapor pressures and boil at lower temperatures.
Module 2: Modeling Matter
Particulate Model of Matter
The particulate model explains the diversity in properties and behaviors of substances based on their internal structure. Matter is composed of a large number of very small particles (atoms, molecules, ions).
Particles: Represent atoms, molecules, or ions.
Scale: Most particles are nanometer-sized ().
Dynamic Nature of Particles
Particles are constantly moving in random directions. Temperature is a measure of the average kinetic energy per particle:
Pressure is determined by the force of particle collisions on container walls:
Interactions and Emergent Properties
Particles interact with each other, and the strength of these interactions depends on the distance between them. Emergent properties, such as density, viscosity, and boiling temperature, arise from collective interactions.
Modeling Gases: Ideal Gas Law
At high temperatures and low pressures, gases can be modeled as non-interacting particles. The ideal gas law relates pressure, volume, temperature, and number of particles:
Where is the Boltzmann constant and is the gas constant.
Phase Transitions: Kinetic vs. Potential Energy
During phase transitions, energy is transferred as potential energy (due to particle interactions), while average kinetic energy per particle remains constant.
Potential Energy (): Decreases as particles move closer due to attractive forces.
Kinetic Energy (): Due to particle motion.
Module 3: Analyzing Particles
Atomic Model of Substances
Substances are composed of particles with different compositions and structures. Atoms are held together by chemical bonds to form molecules.
Elementary Substances: Identical particles made of free or bonded atoms of the same type.
Compounds: Identical particles made of bonded atoms of two or more different types.
Classification of Substances
Type | Composition | Example |
|---|---|---|
Element | Identical atoms | Argon (Ar), Nitrogen (N2) |
Compound | Bonded atoms of different types | Water (H2O), Carbon dioxide (CO2) |
Molecular and Ionic Compounds
Molecular compounds are made of molecules, while ionic compounds are made of ions arranged in lattice networks.
Molecular Formula: Indicates the number and type of atoms in a molecule.
Formula Unit: Represents the proportion of ions in an ionic compound.
Quantitative Analysis: Moles and Molar Mass
The mole is a central unit in chemistry, representing particles (Avogadro's number). Molar mass is the mass of one mole of a substance.
Where is the number of particles, is the number of moles, is the mass, and is the molar mass.
Units of Concentration
Concentration refers to the amount of solute per amount of solvent or solution. Molarity () is defined as:
Module 4: Determining Composition
Experimental Determination of Composition
Experimental techniques such as mass spectrometry and elemental analysis are used to determine the atomic composition of substances.
Mass Spectrometry: Measures mass-to-charge ratio of ions to identify isotopes and molecular fragments.
Elemental Analysis: Determines the percentage of each element in a compound.
Isotopes and Atomic Mass
Isotopes are atoms of the same element with different numbers of neutrons. The average atomic mass is calculated based on the relative abundance of each isotope.
Empirical and Molecular Formulas
The empirical formula gives the simplest integer ratio of atoms in a compound, while the molecular formula gives the actual number of atoms in a molecule.
Steps to Determine Formula:
Calculate mass of each element in a sample.
Convert mass to moles.
Write preliminary formula and reduce to smallest integer subscripts.
Compare empirical formula mass to actual molar mass to determine molecular formula.
Summary
Distinguishing substances relies on identifying differentiating characteristics, understanding phase behavior, and applying the particulate model of matter. Quantitative analysis and experimental techniques allow chemists to determine the composition and properties of substances, which is essential for applications across science and industry.
