Back
BackDynamic Equilibrium and the Equilibrium Constant in Chemical Reactions
Study Guide - Smart Notes

Dynamic Equilibrium in Chemical Reactions
Definition and Characteristics of Dynamic Equilibrium
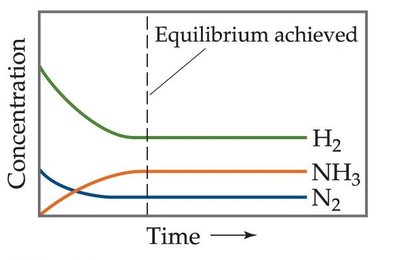
Dynamic equilibrium is a fundamental concept in general chemistry, describing a state in which the rates of the forward and reverse reactions are equal, resulting in constant concentrations of reactants and products. This state is achieved in closed systems and is essential for understanding chemical equilibria.
Dynamic equilibrium: A steady state where forward and reverse processes occur at the same rate.
Concentrations of reactants and products remain constant over time.
Both reactions continue to occur, but there is no net change in concentrations.
Applies to chemical reactions, phase changes, and dissolution processes.
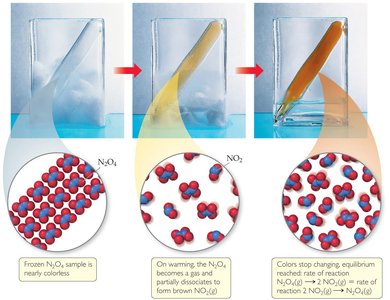
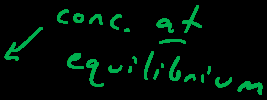
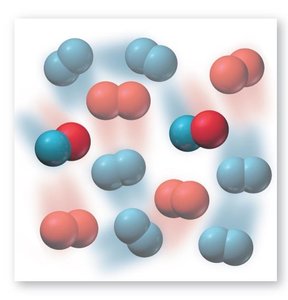
Examples of Dynamic Equilibrium
One classic example is the equilibrium between dinitrogen tetroxide (N2O4) and nitrogen dioxide (NO2):
Reaction:
At equilibrium, the color of the mixture stabilizes, indicating constant concentrations.
The Equilibrium Constant (K)
Law of Mass Action and Equilibrium Constant Expression
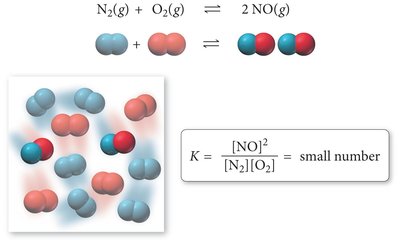
The equilibrium constant quantifies the ratio of product and reactant concentrations at equilibrium. It is derived from the law of mass action and is independent of initial concentrations but dependent on temperature.
Law of Mass Action: For a general reaction , the equilibrium constant is:
The expression is based on stoichiometry, not the reaction mechanism.
K is unitless and reflects the relative concentrations at equilibrium.
Calculating Kc and Kp
For reactions involving gases, equilibrium constants can be expressed in terms of concentrations (Kc) or partial pressures (Kp). The two are related by the ideal gas law.
Kc: Uses molar concentrations.
Kp: Uses partial pressures.
Relationship: , where is the change in moles of gas.
Example: Industrial Nitrogen Fixation (Haber Process)
The synthesis of ammonia is a key industrial equilibrium reaction:
Reaction:
Equilibrium constant expression:

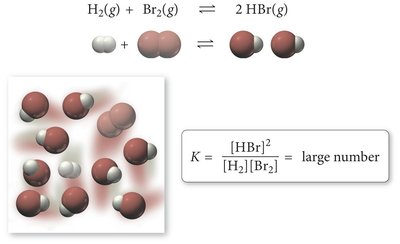
Magnitude and Meaning of the Equilibrium Constant
Interpreting K Values
The magnitude of the equilibrium constant indicates whether a reaction favors products or reactants at equilibrium.
Large K (>1): Reaction favors products; equilibrium lies to the right.
Small K (<1): Reaction favors reactants; equilibrium lies to the left.
K ≈ 1: Significant amounts of both reactants and products are present.
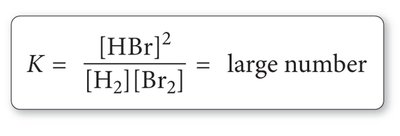
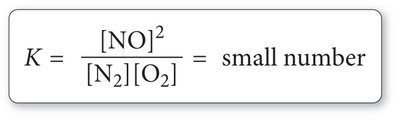
Potential Energy Profiles and Equilibrium
Potential energy diagrams illustrate the energy changes during a reaction and help explain the relationship between activation energy and equilibrium position.
Activation energy (Ea) determines the rate, not the equilibrium position.
ΔE (change in energy) relates to the favorability of products or reactants.
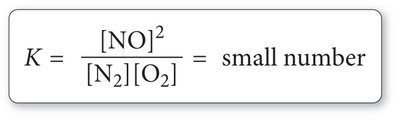
Manipulating Equilibrium Constants
Effect of Stoichiometry and Reaction Direction
The equilibrium constant depends on how the reaction is written. Reversing or scaling the reaction changes the value of K.
Reversing the reaction:
Multiplying the reaction: (where n is the factor)
Combining reactions: Multiply the K values of individual reactions.
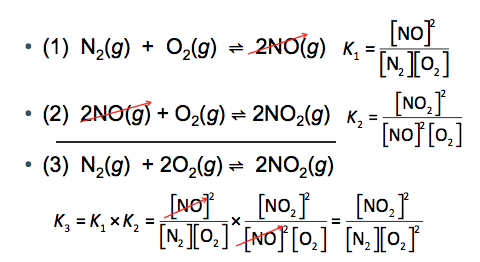
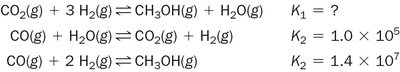
Practice Problems: Calculating and Manipulating K
Practice problems reinforce the calculation and manipulation of equilibrium constants for various reactions.
Write the equilibrium constant expression for each reaction.
Adjust K values for reversed or scaled reactions.
Combine reactions to predict new equilibrium constants.
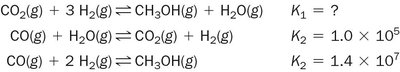
Summary of Key Concepts
Dynamic equilibrium is achieved when forward and reverse rates are equal.
The equilibrium constant (K) describes the ratio of product to reactant concentrations at equilibrium.
K is independent of initial concentrations but dependent on temperature.
The magnitude of K indicates whether a reaction favors products or reactants.
K values can be manipulated algebraically based on reaction stoichiometry and direction.