 Back
BackElectrochemistry and Batteries: Study Notes for General Chemistry
Study Guide - Smart Notes

Electrochemistry and Batteries
Introduction to Electrochemistry
Electrochemistry is the study of chemical processes that involve the movement of electrons, resulting in the generation of electric current or the use of electricity to drive nonspontaneous reactions. This field is essential for understanding batteries, fuel cells, and electrolysis.
Cell Potentials, Gibbs Free Energy, and Equilibrium
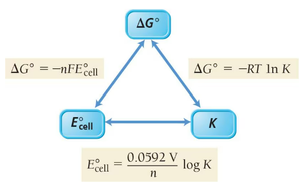
Relationship Between ΔG, Eocell, and K
The spontaneity of an electrochemical reaction is determined by the cell potential (Ecell), the Gibbs free energy change (ΔG), and the equilibrium constant (K). These quantities are interrelated as follows:
ΔG < 0: The reaction is spontaneous.
Ecell > 0: The cell can do electrical work spontaneously.
K > 1: Products are favored at equilibrium.
The following equations summarize these relationships:
Standard Reduction Potentials
Standard reduction potentials (Eo) are measured under standard conditions (1 M, 1 atm, 25°C) and are used to predict the direction of electron flow in electrochemical cells. The more positive the Eo, the greater the tendency to be reduced.
Half-Reaction | Eo (V) |
|---|---|
O2(g) + 4 H+(aq) + 4 e- → 2 H2O(l) | 1.23 |
MnO2(s) + 4 H+(aq) + 2 e- → Mn2+(aq) + 2 H2O(l) | 1.21 |
Br2(l) + 2 e- → 2 Br-(aq) | 1.09 |
Ag+(aq) + e- → Ag(s) | 0.80 |
Cu2+(aq) + 2 e- → Cu(s) | 0.34 |
Zn2+(aq) + 2 e- → Zn(s) | -0.76 |
Al3+(aq) + 3 e- → Al(s) | -1.66 |
Mg2+(aq) + 2 e- → Mg(s) | -2.37 |


Calculating Cell Potentials
Standard Cell Potential (Eocell)
The standard cell potential is calculated from the standard reduction potentials of the cathode and anode:
To determine the cell potential for a given pair of half-reactions, identify which is the reduction (cathode) and which is the oxidation (anode) reaction.
Nonstandard Conditions: The Nernst Equation
When concentrations are not 1 M, the Nernst equation is used to calculate the cell potential:
At 25°C (298 K):
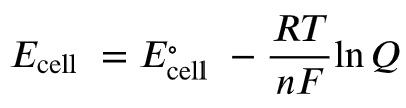
Where Q is the reaction quotient, n is the number of electrons transferred, R is the gas constant, T is temperature in Kelvin, and F is Faraday's constant.
Electrochemical Cells and Batteries
Galvanic (Voltaic) Cells
Galvanic cells use spontaneous redox reactions to generate electricity. The cell is constructed with two half-cells connected by a salt bridge, allowing ion flow to maintain charge balance.
Concentration Cells
Concentration cells are a special type of galvanic cell where both electrodes are made of the same material, but the ion concentrations differ. The cell generates a potential due to the concentration gradient.
As the cell operates, the concentrations tend to equalize, and the cell potential decreases.
Example calculation for a Cu concentration cell:
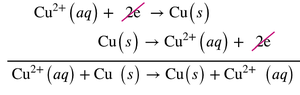
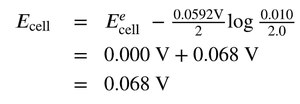
Electrochemistry in Biological Systems
Cells in the human body use ion gradients (such as Na+ and K+) across membranes to generate electrical potentials, essential for nerve impulses and muscle contraction.
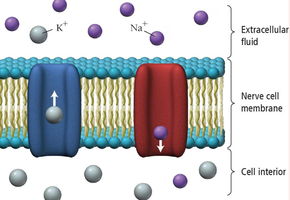
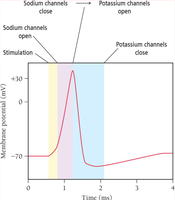
Types of Batteries
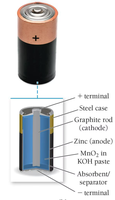
Dry Cell (Alkaline Battery)
Dry cells use Zn/MnO2 chemistry, are non-rechargeable, and produce about 1.5 V per cell. They are widely used in household electronics.
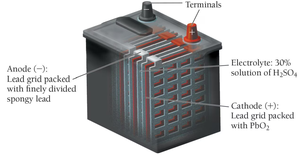
Lead-Acid Storage Battery
Lead-acid batteries use Pb and PbO2 electrodes in sulfuric acid. They are rechargeable and commonly used in automobiles, producing about 2 V per cell (6 cells for 12 V).
Nickel-Cadmium (NiCad) and Nickel-Metal Hydride (NiMH) Batteries
NiCad batteries use Ni/Cd chemistry, are rechargeable, and produce about 1.3 V per cell. NiMH batteries use a metal hydride instead of cadmium, are lighter, and have variable voltage.
Lithium-Ion Batteries
Lithium-ion batteries use lithium and transition metal oxides. They are lightweight, rechargeable, and have high energy density, but require careful handling due to fire risk.
Fuel Cells
Fuel cells convert chemical energy from fuels (like H2 or ethanol) directly into electricity. They require a continuous supply of reactants and are used in specialized applications such as space shuttles and breathalyzers.
Electrolytic Cells
Electrolytic cells use electricity to drive nonspontaneous chemical reactions. Applications include water electrolysis, metal plating, and industrial gas generation.
Summary Table: Key Battery Types
Battery Type | Chemistry | Voltage (per cell) | Rechargeable? | Notes |
|---|---|---|---|---|
Dry Cell | Zn/MnO2 | ~1.5 V | No | Common, non-rechargeable |
Lead-Acid | Pb/PbO2 in H2SO4 | ~2 V | Yes | Automotive, heavy |
NiCad | Ni/Cd | ~1.3 V | Yes | Rechargeable, toxic Cd |
NiMH | Ni/Metal Hydride | Variable | Yes | Lighter, less toxic |
Li-Ion | Li/Transition Metal | Variable | Yes | High energy, fire risk |
Fuel Cell | H2/O2, EtOH/O2 | Variable | No | Continuous fuel supply |
Key Equations and Constants
Faraday's constant (F): 96,485 C/mol e-
Gas constant (R): 8.314 J/(mol·K)
Standard temperature: 298 K (25°C)
Practice Problems
Calculate ΔG and K for a given cell reaction using the equations above.
Balance redox reactions in acidic and basic conditions.
Design and label an electrochemical cell using standard notation.
Additional info: The notes above integrate and expand upon the provided lecture slides, images, and tables, ensuring a comprehensive and academically rigorous summary suitable for general chemistry students.
