 Back
BackElectrochemistry and Redox Reactions: Study Notes for General Chemistry
Study Guide - Smart Notes

Electrochemistry and Redox Reactions
Introduction to Electrochemistry
Electrochemistry is the study of chemical processes that involve the movement of electrons, known as redox (reduction-oxidation) reactions. These reactions are fundamental to understanding batteries, corrosion, and many biological processes. In electrochemical cells, chemical energy is converted to electrical energy or vice versa.
Thermodynamics and Electrochemistry
Gibbs Free Energy and Spontaneity
The spontaneity of a chemical reaction is determined by the change in Gibbs free energy (ΔG). A negative ΔG indicates a spontaneous process, while a positive ΔG indicates a nonspontaneous process. The relationship between ΔG and the equilibrium constant (K) is crucial for predicting reaction direction and extent.
Key Equation:
Standard State: For gases, 1 bar pressure; for solutions, 1 M concentration; for pure solids and liquids, the pure substance.
Example: Calculating ΔG for a target reaction using steps and their respective ΔG values.
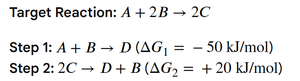
Equilibrium and Free Energy
At equilibrium, ΔG = 0, and the relationship between standard free energy change and the equilibrium constant is given by:
Where R is the gas constant (8.314 J/mol·K) and T is temperature in Kelvin.

Redox Reactions and Oxidation States
Oxidation and Reduction
Redox reactions involve the transfer of electrons between species. Oxidation is the loss of electrons, while reduction is the gain of electrons. These processes always occur together in a chemical reaction.
Oxidation: Increase in oxidation state (loss of electrons).
Reduction: Decrease in oxidation state (gain of electrons).
Example: The reaction of sodium and chlorine to form sodium chloride involves sodium being oxidized and chlorine being reduced.
Assigning Oxidation States
Oxidation states are used to keep track of electron transfer in redox reactions. The following rules are used to assign oxidation states:
The oxidation state of an atom in its elemental form is 0.
The oxidation state of a monatomic ion is equal to its charge.
The sum of oxidation states in a neutral molecule is 0; in a polyatomic ion, it equals the ion's charge.
Metals in compounds have positive oxidation states (Group 1A: +1, Group 2A: +2).
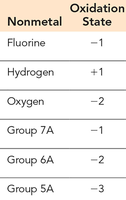
Nonmetals follow a hierarchy (see table below).
Nonmetal | Oxidation State |
|---|---|
Fluorine | -1 |
Hydrogen | +1 |
Oxygen | -2 |
Group 7A | -1 |
Group 6A | -2 |
Group 5A | -3 |
Examples of Assigning Oxidation States
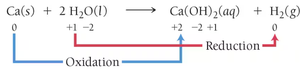
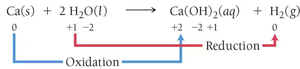
Practice assigning oxidation numbers to elements in compounds and ions. For example, in Ca(s) + 2 H2O(l) → Ca(OH)2(aq) + H2(g), calcium is oxidized and hydrogen is reduced.
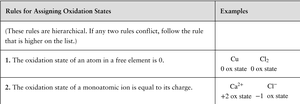
Rules for Assigning Oxidation States (Summary Table)
Rule | Examples |
|---|---|
1. Elemental form: 0 | Cu: 0, Cl2: 0 |
2. Monatomic ion: charge | Ca2+: +2, Cl-: -1 |
3. Sum in molecule/ion | H2O: 2(+1) + (-2) = 0 |
4. Metals in compounds | NaCl: +1, CaF2: +2 |


Balancing Redox Reactions
Balancing in Acidic and Basic Solution
Redox reactions are balanced by separating them into half-reactions, balancing mass and charge, and then combining them. In acidic solution, H+ and H2O are used; in basic solution, OH- is used to neutralize protons.
Assign oxidation states to identify oxidized and reduced species.
Write and balance half-reactions for mass and charge.
Balance O with H2O, H with H+ (acid) or OH- (base).
Combine half-reactions, ensuring electrons cancel.
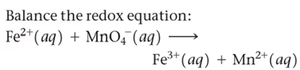
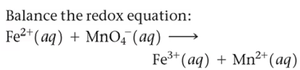
Electrochemical Cells
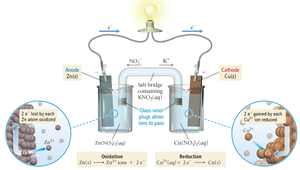
Galvanic (Voltaic) and Electrolytic Cells
Electrochemical cells are devices that convert chemical energy to electrical energy (galvanic/voltaic) or use electrical energy to drive nonspontaneous reactions (electrolytic). Both types have two electrodes: anode (oxidation) and cathode (reduction).
Galvanic Cell: Spontaneous reaction, generates electricity.
Electrolytic Cell: Nonspontaneous reaction, requires external voltage.
Electron flow: always from anode to cathode.
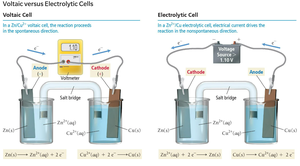
Cell Notation
Cell notation is a shorthand for representing electrochemical cells. The anode (where oxidation occurs) is written on the left, and the cathode (where reduction occurs) on the right. A single vertical line (|) separates different phases, and a double vertical line (||) represents the salt bridge.
General format: Anode (reactant | product) || Cathode (reactant | product)
Platinum electrode is used when reactants or products are not solids.
Standard Electrode Potentials
Reference Electrode and Standard Potentials
The standard hydrogen electrode (SHE) is assigned a potential of 0 V and is used as a reference to measure other half-cell potentials. The standard cell potential (Ecell0) is calculated from the difference between the cathode and anode potentials.
Key Equation:
Positive Ecell0 indicates a spontaneous reaction.
Predicting Spontaneity and Battery Chemistry
Spontaneous redox reactions have a positive cell potential. The relative positions of half-reactions in a standard reduction potential table can be used to predict which reactions will occur spontaneously.
Any reduction half-reaction is spontaneous when paired with the reverse of a half-reaction below it in the table.
Metals above hydrogen in the activity series dissolve in acid; those below do not.
Quantifying Batteries
Current, Charge, and Potential
Electrochemical cells generate or use electrical current, which is the flow of electrons. The cell potential (voltage) is a measure of the driving force for electron flow.
Current (I): Measured in amperes (A), where 1 A = 1 coulomb/second.
Charge: 1 coulomb = 6.242 × 1018 electrons.
Potential (V): 1 V = 1 joule/coulomb.
Cell Potential (Ecell): Difference in potential between two half-cells.
Summary
Electrochemistry connects chemical reactions to electrical energy through redox processes. Understanding oxidation states, balancing redox reactions, and calculating cell potentials are essential skills in general chemistry. These concepts are foundational for batteries, corrosion, and many industrial and biological processes.
