 Back
BackElectrochemistry and Redox Reactions: Study Notes for General Chemistry
Study Guide - Smart Notes

Electrochemistry and Redox Reactions
Oxidation-Reduction Reactions
Oxidation-reduction (redox) reactions are fundamental to electrochemistry, involving the transfer of electrons between chemical species. These reactions can be broken down into two half-reactions: one for oxidation (loss of electrons) and one for reduction (gain of electrons).
Oxidation: Loss of electrons by a species; increase in oxidation number.
Reduction: Gain of electrons by a species; decrease in oxidation number.
Oxidizing Agent: Causes another species to be oxidized; itself is reduced.
Reducing Agent: Causes another species to be reduced; itself is oxidized.
Mnemonic: "LEO the lion says GER" (Loss of Electrons is Oxidation; Gain of Electrons is Reduction) and "OIL RIG" (Oxidation Is Loss, Reduction Is Gain).
Example: The reaction between sodium and chlorine:
Oxidation half-reaction:
Reduction half-reaction:
Overall reaction:
Assigning Oxidation Numbers
Oxidation numbers are used to keep track of electron transfer in reactions. Proper assignment is essential for identifying redox processes.
Elements: Oxidation number is zero.
Monatomic ions: Oxidation number equals the ion's charge.
Oxygen: Usually -2; -1 in peroxides.
Hydrogen: +1 in most compounds; -1 in metal hydrides.
Halogens: Fluorine is always -1; other halogens are -1 except when combined with oxygen or more electronegative halogens.
Compounds: Sum of oxidation numbers is zero; for polyatomic ions, sum equals the ion's charge.
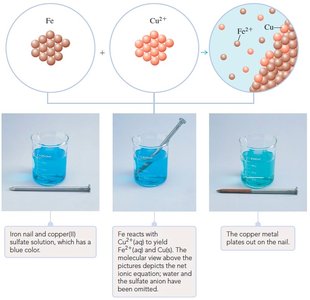
Example: Assigning oxidation numbers in Fe(s) + CuSO4(aq) → FeSO4(aq) + Cu(s):
Fe(s): 0
Cu2+(aq): +2
Fe2+(aq): +2
Cu(s): 0
Types of Redox Reactions
Redox reactions can be classified into several types based on their reactants and products:
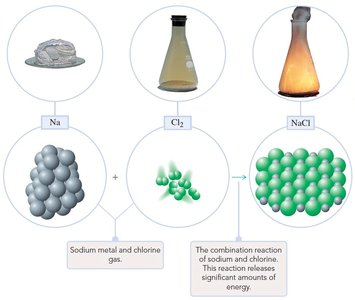
Combination Reaction: Two substances combine to form a third. Example:
Decomposition Reaction: A single compound breaks down into two or more substances. Example:
Displacement Reaction: An element displaces another from a compound. Example:
Combustion Reaction: A substance reacts with oxygen, releasing heat and producing a flame. Example:
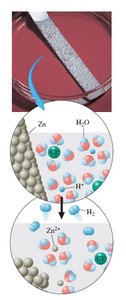
Activity Series of the Elements
The activity series ranks elements by their tendency to lose electrons (be oxidized). Elements at the top are strong reducing agents and react vigorously, while those at the bottom are weak reducing agents.
Top of series: React vigorously with acids and water (e.g., Li, K, Ba).
Middle: React with acids to give H2 (e.g., Mg, Zn, Fe).
Bottom: Do not react with acids to give H2 (e.g., Cu, Ag, Au).
Reactivity | Elements |
|---|---|
React vigorously with acids/water | Li, K, Ba, Ca, Na |
React with acids | Mg, Al, Zn, Cr, Fe, Cd, Co, Ni, Sn, Pb |
Do not react with acids | H2, Cu, Hg, Ag, Au |
Balancing Redox Reactions: The Half-Reaction Method
Balancing redox reactions involves separating the process into two half-reactions, balancing atoms and charges, and then combining them.
Assign oxidation numbers to identify what is oxidized and reduced.
Write unbalanced half-reactions for oxidation and reduction.
Balance atoms and charges by adding electrons, H+ (acidic), or OH- (basic), and H2O as needed.
Scale half-reactions so electrons cancel when added together.
Add the half-reactions to obtain the balanced overall equation.
Example:
Oxidation:
Reduction:
Combined:
Electrochemical Cells
Voltaic (Galvanic) Cells
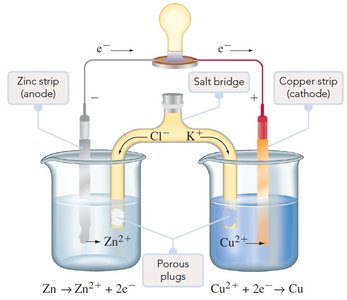
A voltaic cell is an electrochemical cell in which a spontaneous redox reaction generates an electric current. It consists of two half-cells connected by an external circuit and a salt bridge.
Electrochemical cell: System with electrodes in an electrolyte, where chemical reactions generate or use electric current.
Voltaic (galvanic) cell: Spontaneous reaction generates current.
Electrolytic cell: External energy drives a nonspontaneous reaction.
Half-cell: Portion where a half-reaction occurs (oxidation or reduction).
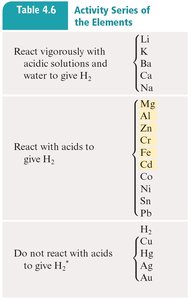
Example: Zn(s) loses electrons (oxidized), Cu2+(aq) gains electrons (reduced), electrons flow from Zn to Cu through the external circuit.
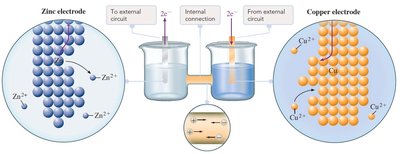
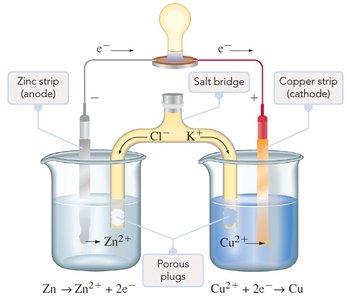
Salt Bridge and Electron Flow
The salt bridge is essential for maintaining charge balance by allowing ion flow between half-cells without mixing solutions. In a voltaic cell:
Anode: Site of oxidation; electrons are released and flow to the external circuit.
Cathode: Site of reduction; electrons enter from the external circuit.
Salt bridge: Cations move toward the cathode, anions toward the anode, maintaining electroneutrality.
Electron flow: From anode (negative) to cathode (positive).
Summary Table: Voltaic Cell Components
Component | Function |
|---|---|
Anode | Oxidation occurs; electrons released |
Cathode | Reduction occurs; electrons accepted |
Salt Bridge | Maintains charge balance by ion flow |
External Circuit | Electrons flow from anode to cathode |
Check Your Understanding: Voltaic Cell Construction
When constructing a voltaic cell with copper and aluminum electrodes, the aluminum is oxidized (anode), and copper is reduced (cathode). Electrons flow from aluminum to copper through the external circuit.
Additional info: These notes cover the essential concepts of redox reactions, oxidation numbers, types of redox reactions, activity series, balancing redox equations, and the structure and function of electrochemical cells, as outlined in general chemistry curriculum chapters 4 and 19.
