 Back
BackElectrochemistry: Principles, Cells, and Applications
Study Guide - Smart Notes

Electrochemistry: Principles and Applications
Introduction to Electrochemistry
Electrochemistry is the branch of chemistry that studies the relationship between electrical energy and chemical reactions, particularly those involving electron transfer (redox reactions). It encompasses both spontaneous and nonspontaneous processes, including the generation of electricity from chemical reactions (as in batteries) and the use of electricity to drive chemical changes (as in electrolysis).
Galvanic (Voltaic) Cells: Use chemical reactions to produce electricity (e.g., batteries).
Electrolytic Cells: Use electricity to drive nonspontaneous chemical reactions (e.g., electroplating, aluminum production).
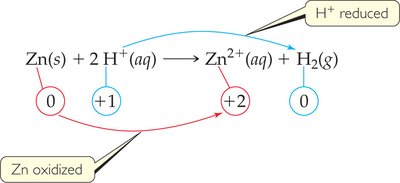
Redox Reactions and Oxidation Numbers
Assigning Oxidation Numbers
Oxidation numbers are used to keep track of electron transfer in redox reactions. The rules for assigning oxidation numbers are:
Elements in their pure form: 0
Monatomic ions: Equal to their charge
Fluorine: –1
Oxygen: –2 (except in peroxides, where it is –1)
Hydrogen: +1 (except in metal hydrides, where it is –1)
The sum of oxidation numbers in a compound is 0; in a polyatomic ion, it equals the ion's charge.
Identifying Oxidation and Reduction
In a redox reaction:
Oxidation: Increase in oxidation number (loss of electrons)
Reduction: Decrease in oxidation number (gain of electrons)
The substance that is reduced is the oxidizing agent; the substance that is oxidized is the reducing agent.
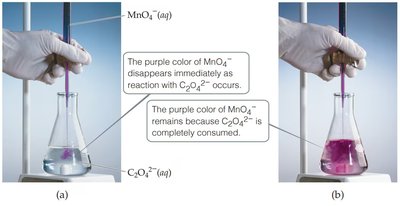
Visualizing Redox Reactions
Color changes can indicate redox reactions, such as the reaction between permanganate (MnO4–) and oxalate (C2O42–), where the purple color of MnO4– disappears as it is reduced.
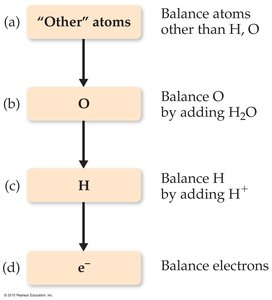
Balancing Redox Equations: The Half-Reaction Method
Steps for Balancing Redox Equations
The half-reaction method is a systematic approach to balancing redox equations, especially in aqueous solution:
Write separate half-reactions for oxidation and reduction.
Balance all elements except H and O.
Balance O by adding H2O.
Balance H by adding H+.
Balance charge by adding electrons (e–).
Multiply half-reactions by appropriate factors to equalize electrons.
Add the half-reactions and simplify.
Check that both mass and charge are balanced.
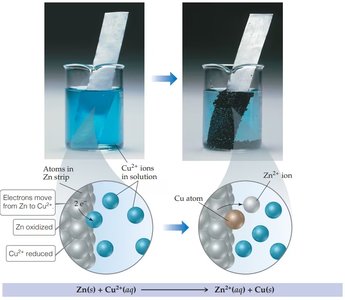
Electrochemical Cells
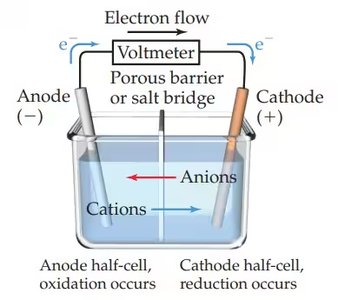
Galvanic (Voltaic) Cells
Galvanic cells convert chemical energy into electrical energy through spontaneous redox reactions. The two half-reactions are separated into different compartments, allowing electrons to flow through an external circuit.
Anode: Site of oxidation (loss of electrons)
Cathode: Site of reduction (gain of electrons)
Salt Bridge: Maintains electrical neutrality by allowing ion flow between compartments

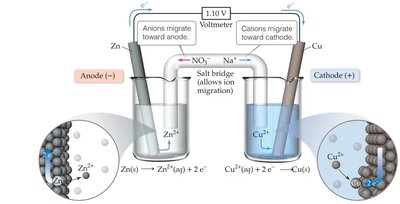
Mnemonic for Electrodes
Remember: Oxidation occurs at the Anode, Reduction at the Cathode ("Red Cat, An Ox").
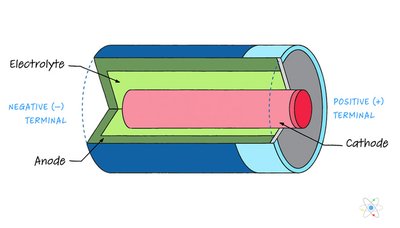
Structure of a Battery
Batteries are practical applications of galvanic cells, with an anode, cathode, and electrolyte facilitating electron flow.
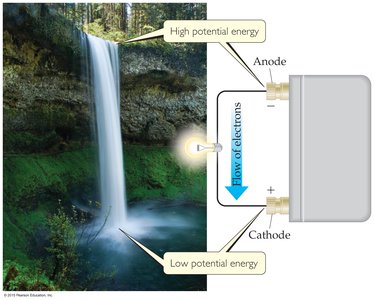
Cell Potential and Electromotive Force (EMF)
Definition and Measurement
The cell potential (Ecell) or electromotive force (emf) is the voltage difference between the two electrodes of an electrochemical cell. It is measured in volts (V), where 1 V = 1 J/C (joule per coulomb).
Standard Cell Potentials
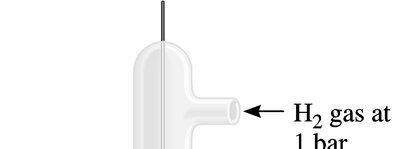
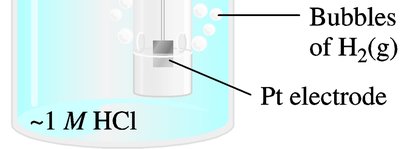
Standard cell potentials (Eo) are measured under standard conditions: 1 M concentrations, 1 bar (or 1 atm) pressure, and 25°C (298 K). The standard hydrogen electrode (SHE) is used as the reference, assigned a potential of 0 V.

Standard Reduction Potentials Table
Standard reduction potentials are tabulated for various half-reactions. The more positive the Eo, the greater the tendency to be reduced (act as an oxidant).
Ered (V) | Reduction Half-Reaction |
|---|---|
+2.87 | F2(g) + 2 e– → 2 F–(aq) |
+1.51 | MnO4–(aq) + 8 H+(aq) + 5 e– → Mn2+(aq) + 4 H2O(l) |
+1.36 | Cl2(g) + 2 e– → 2 Cl–(aq) |
+0.34 | Cu2+(aq) + 2 e– → Cu(s) |
0.00 | 2 H+(aq) + 2 e– → H2(g) |
–0.76 | Zn2+(aq) + 2 e– → Zn(s) |
–2.71 | Na+(aq) + e– → Na(s) |
–3.05 | Li+(aq) + e– → Li(s) |

Calculating Cell Potentials
The standard cell potential is calculated as:
Where both values are standard reduction potentials. The more positive Eo is the cathode.
Thermodynamics and Electrochemistry
Relationship to Free Energy and Equilibrium
Electrochemical cell potentials are directly related to thermodynamic quantities:
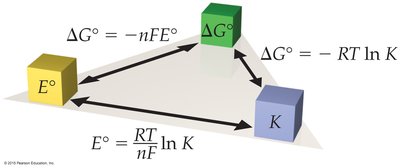
Where:
n = number of moles of electrons transferred
F = Faraday's constant (96,485 C/mol e–)
R = gas constant (8.314 J/mol·K)
T = temperature in Kelvin
K = equilibrium constant
The Nernst Equation
The Nernst equation allows calculation of cell potential under non-standard conditions:
Where Q is the reaction quotient. At 25°C, this simplifies to:
Electrolysis and Faraday’s Law
Electrolysis
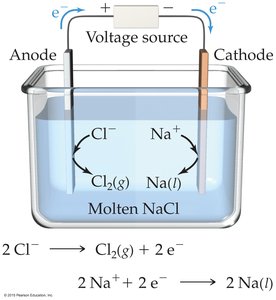
Electrolysis uses electrical energy to drive nonspontaneous chemical reactions. Examples include the decomposition of molten NaCl and electroplating of metals.
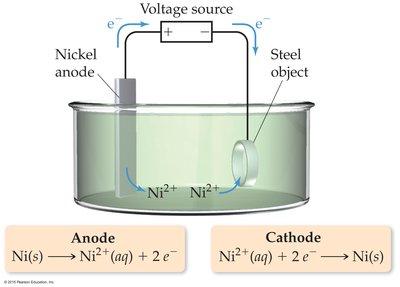
Faraday’s Law of Electrolysis
Faraday’s Law states that the amount of substance produced at each electrode is directly proportional to the quantity of charge passed through the cell.
Faraday’s constant (F): 96,485 C/mol e–
Calculation:

Summary Table: Key Concepts in Electrochemistry
Concept | Description |
|---|---|
Oxidation | Loss of electrons; occurs at the anode |
Reduction | Gain of electrons; occurs at the cathode |
Galvanic Cell | Spontaneous redox reaction produces electricity |
Electrolytic Cell | Electricity drives a nonspontaneous reaction |
Cell Potential (Ecell) | Measured in volts; difference in potential between electrodes |
Faraday’s Law | Relates charge passed to amount of substance produced |
Additional info: The above notes include expanded academic context and examples for clarity and completeness, as well as logical grouping of fragmented topics for a cohesive study guide.
