 Back
BackElectrochemistry: Redox Reactions, Galvanic and Electrolytic Cells, and Applications
Study Guide - Smart Notes

Electrochemistry
Introduction to Electrochemistry
Electrochemistry is the branch of chemistry that studies the relationship between electricity and chemical reactions, particularly those involving electron transfer (redox reactions). It is fundamental to understanding batteries, corrosion, electrolysis, and many industrial processes.
Redox Basics & Agents
Oxidation and Reduction
Redox (reduction-oxidation) reactions involve the transfer of electrons between chemical species. The substance that loses electrons is oxidized, while the substance that gains electrons is reduced.
Oxidation: Loss of electrons; oxidation state becomes more positive or less negative.
Reduction: Gain of electrons; oxidation state becomes more negative or less positive.
Oxidizing Agent: The species that causes oxidation by accepting electrons (itself is reduced).
Reducing Agent: The species that causes reduction by donating electrons (itself is oxidized).

Examples of Redox Reactions in Industry
Claus Process: Used to recover sulfur from natural gas and crude oil. The overall reaction is:
Haber Process: Synthesis of ammonia for fertilizers and explosives:

Identifying Oxidation and Reduction in Reactions
To determine which element is oxidized or reduced, compare oxidation states before and after the reaction. The oxidizing agent is reduced, and the reducing agent is oxidized.
Example: - Iron is oxidized (Fe2+ to Fe3+), manganese is reduced (MnO4- to Mn2+).
Oxidizing and Reducing Agents in Everyday Substances
Common oxidizing agents include O2, Cl2, and KMnO4. Reducing agents include H2, metals like Zn, and organic compounds like gasoline.
Changes in Oxidation State
Recognizing Redox Reactions
Redox reactions are characterized by changes in oxidation states. Combustion, rusting, and reactions involving pure elements are always redox processes.
Combustion:
Rusting:
Balancing Aqueous-phase Redox Equations
Steps for Balancing Redox Reactions
Redox reactions in aqueous solution are balanced by separating them into half-reactions, balancing atoms and charges, and then combining them.
Write oxidation and reduction half-reactions.
Balance all elements except H and O.
Balance O by adding H2O, H by adding H+ (in acidic solution), and charge by adding electrons.
Multiply half-reactions to equalize electrons and add together.
Galvanic (Voltaic) Cells
Structure and Function
Galvanic cells convert chemical energy into electrical energy through spontaneous redox reactions. They consist of two half-cells connected by a salt bridge and an external circuit.
Anode: Site of oxidation (loses electrons).
Cathode: Site of reduction (gains electrons).
Salt Bridge: Maintains electrical neutrality by allowing ion flow.
Electron Flow: From anode to cathode through the external wire.
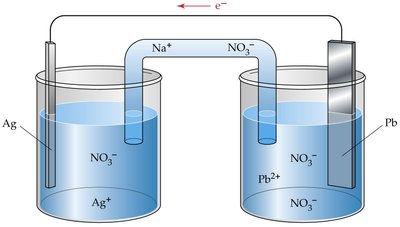
Cell Notation and Calculations
Cell notation summarizes the components of a galvanic cell. The standard cell potential () is calculated from standard reduction potentials:

Standard Reduction Potentials
Definition and Use
Standard reduction potentials () measure the tendency of a species to gain electrons under standard conditions (1 M, 1 atm, 298 K). The more positive the $E^\circ$, the greater the tendency to be reduced.
Standard hydrogen electrode (SHE) is assigned V.
Tables of values are used to predict cell voltages and spontaneity.

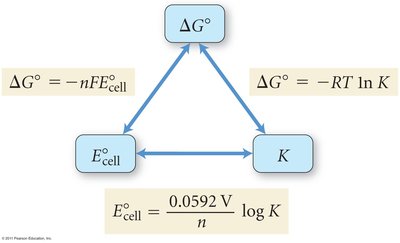
Relationship Between ΔG°, K, and E°cell
Thermodynamic Connections
The spontaneity of electrochemical reactions is related to the standard cell potential, equilibrium constant, and standard free energy change:
(at 25°C)
Nernst Equation and Non-Standard Cells
Nernst Equation
The Nernst equation allows calculation of cell potential under non-standard conditions:
At 25°C:
Q is the reaction quotient.
Corrosion
Mechanism of Corrosion
Corrosion is the deterioration of metals by redox reactions with environmental agents, such as oxygen and water. Iron rusting is a common example, involving the oxidation of Fe to Fe2+ and subsequent reactions to form hydrated iron(III) oxide (rust).
Electrolytic Cells and Faraday’s Law
Electrolytic Cells
Electrolytic cells use electrical energy to drive non-spontaneous redox reactions. They are used in electroplating, metal purification, and industrial processes like the chloralkali process.
Cathode: Site of reduction (gains electrons).
Anode: Site of oxidation (loses electrons).
Faraday’s Law of Electrolysis
Faraday’s Law relates the amount of substance produced at an electrode to the quantity of electricity passed through the cell:
I = current (A), t = time (s), n = number of electrons, = Faraday’s constant (96500 C/mol e-).
Applications and Industrial Processes
Industrial Electrochemistry
Chloralkali Process: Electrolysis of brine to produce Cl2, H2, and NaOH.
Aluminum Production: Electrolysis of molten Al2O3 to obtain pure aluminum metal.
Summary Table: Key Electrochemical Concepts
Concept | Definition | Key Equation |
|---|---|---|
Oxidation | Loss of electrons | — |
Reduction | Gain of electrons | — |
Cell Potential () | Voltage produced by a cell | |
Gibbs Free Energy () | Spontaneity of reaction | |
Nernst Equation | Cell potential under non-standard conditions | |
Faraday’s Law | Amount of substance produced in electrolysis |
