 Back
BackElectrochemistry: Redox Reactions, Voltaic Cells, and Applications
Study Guide - Smart Notes

Electrochemistry
Introduction to Electrochemistry
Electrochemistry is the study of chemical processes that involve the movement of electrons, known as redox (reduction-oxidation) reactions. These reactions are fundamental to understanding batteries, corrosion, and many industrial processes.
Balancing Oxidation-Reduction (Redox) Reactions
Assigning Oxidation Numbers
Oxidation numbers (or states) are used to keep track of electron transfer in redox reactions. The rules for assigning oxidation numbers are hierarchical and must be followed in order:
The oxidation state of an atom in a free element is zero (e.g., He: 0, H2: 0).
The oxidation state of a monoatomic ion equals its charge (e.g., Na+: +1, Cl–: –1).
The sum of oxidation states in a compound equals the compound's charge (e.g., H2O: 0, NO3–: –1).
Group 1A metals: +1; Group 2A metals: +2 in compounds.
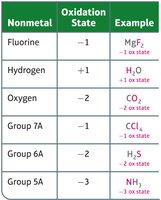
Nonmetals have typical oxidation states as shown below:
Nonmetal | Oxidation State | Example |
|---|---|---|
Fluorine | –1 | MgF2 |
Hydrogen | +1 | H2O |
Oxygen | –2 | CO2 |
Group 7A | –1 | CCl4 |
Group 6A | –2 | H2S |
Group 5A | –3 | NH3 |
Balancing Redox Reactions
Redox reactions must be balanced for both mass and charge. The half-reaction method is commonly used, especially for reactions in aqueous solutions:
Assign oxidation states to all atoms; identify what is oxidized and reduced.
Separate the reaction into two half-reactions (oxidation and reduction).
Balance all elements except H and O.
Balance O by adding H2O.
Balance H by adding H+ (for acidic solutions).
Balance charge by adding electrons.
Multiply half-reactions to equalize electrons, then add together.
Verify mass and charge balance.
For basic solutions, after adding H+, add OH– to both sides to neutralize H+ and form water.
Redox Terminology and Memory Aids
Definitions and Mnemonics
Oxidation: Loss of electrons (OIL: Oxidation Is Loss).
Reduction: Gain of electrons (RIG: Reduction Is Gain).
LEO the lion says GER: Lose Electrons = Oxidation, Gain Electrons = Reduction.

Voltaic (Galvanic) Cells
Structure and Function
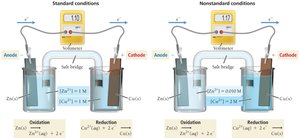
A voltaic cell is an electrochemical cell that generates electrical energy from a spontaneous redox reaction. It consists of two half-cells connected by a salt bridge, with each half-cell containing an electrode and an electrolyte. The anode is where oxidation occurs (negative sign), and the cathode is where reduction occurs (positive sign). Electrons flow from anode to cathode through an external circuit.
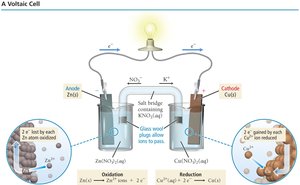
Cell Potential (Ecell)
The cell potential, or electromotive force (emf), is the voltage produced by the cell. It is calculated as:
Standard cell potentials (E°cell) are measured under standard conditions (1 M, 1 atm, 25°C).
Batteries
Types of Batteries and Their Chemistry
Batteries are practical applications of voltaic cells. They use redox reactions to provide portable electrical energy. Common types include:
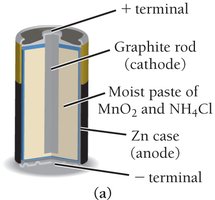
Dry Cell (Zinc-Carbon): Anode is zinc, cathode is a carbon rod surrounded by MnO2. Electrolyte is a moist paste of MnO2 and NH4Cl.
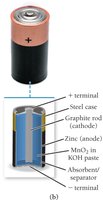
Alkaline Battery: Similar to dry cell but uses KOH as the electrolyte and a steel case. Higher capacity and longer life.
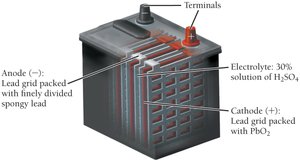
Lead-Acid Battery: Used in cars; anode is lead, cathode is lead(IV) oxide, and the electrolyte is sulfuric acid. Rechargeable and provides high current.
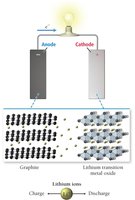
Lithium-Ion Battery: Used in modern electronics; anode is graphite, cathode is a lithium metal oxide. High energy density and rechargeable.
Fuel Cell: Converts chemical energy from fuel (e.g., H2 and O2) directly into electricity. Environmentally friendly, with water as the only byproduct.
Corrosion
Mechanism and Prevention
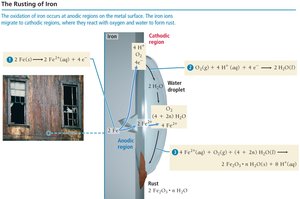
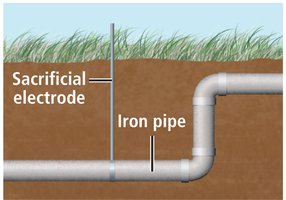
Corrosion is the unwanted oxidation of metals, often due to environmental exposure to oxygen and water. Rusting of iron is a common example, requiring both water and oxygen. Some metals, like aluminum, form protective oxide layers, while others, like iron, degrade.
Prevention methods include painting, galvanization (coating with zinc), and using sacrificial electrodes.

Standard Electrode Potentials
Reference Electrode and Table Use
Standard electrode potentials are measured relative to the Standard Hydrogen Electrode (SHE), which is assigned a potential of 0.00 V. The table of standard reduction potentials allows prediction of cell voltages and spontaneity of reactions.
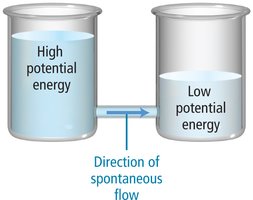

Predicting Reaction Direction
Electrons flow from the half-cell with more negative potential (higher energy) to the one with more positive potential (lower energy). A positive E°cell indicates a spontaneous reaction.
Mnemonic: N.I.O. (Negative Is Oxidation), P.I.R. (Positive Is Reduction).
Free Energy and Equilibrium Constant
Relationship to Cell Potential
The cell potential is directly related to the Gibbs free energy change (ΔG) and the equilibrium constant (K):
(at 25°C)
Where n is the number of moles of electrons transferred, and F is the Faraday constant.
Nonstandard Conditions: The Nernst Equation
Effect of Concentration on Cell Potential
Under nonstandard conditions, the cell potential is affected by the concentrations of reactants and products. The Nernst equation is used to calculate the cell potential:
Where Q is the reaction quotient.
Electrolytic Cells
Driving Nonspontaneous Reactions
Electrolytic cells use electrical energy to drive nonspontaneous chemical reactions. Electron flow is still from anode to cathode, but the cell requires an external voltage source. Applications include water electrolysis and electroplating.
Electrolysis of Water: Splits water into hydrogen and oxygen gases.
Electroplating: Deposits a thin layer of metal onto another material for protection or aesthetics.
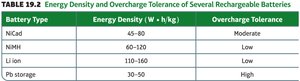
Summary Table: Key Battery Types
Battery Type | Energy Density (W·h/kg) | Overcharge Tolerance |
|---|---|---|
NiCd | 45–80 | Moderate |
NiMH | 60–120 | Low |
Li Ion | 110–160 | Low |
Pb storage | 30–50 | High |
Additional info: This guide covers the essential concepts of electrochemistry, including redox reactions, voltaic and electrolytic cells, batteries, corrosion, and the relationship between cell potential, free energy, and equilibrium. For further practice, work through example problems involving balancing redox reactions, calculating cell potentials, and applying the Nernst equation.
