 Back
BackElectrochemistry: Voltaic Cells, EMF, Standard Electrode Potentials, and Redox Agents
Study Guide - Smart Notes

Electrochemistry: Voltaic Cells, EMF, Standard Electrode Potentials, and Redox Agents
Introduction to Electrochemistry
Electrochemistry is the study of chemical processes that involve the movement of electrons, resulting in the generation of electric current. This field is fundamental to understanding how batteries, fuel cells, and other devices operate. The following notes cover key concepts in Chapter 18: Electrochemistry, focusing on voltaic cells, cell electromotive force (EMF), standard electrode potentials, and the identification of oxidizing and reducing agents.
Voltaic Cells
Definition and Structure
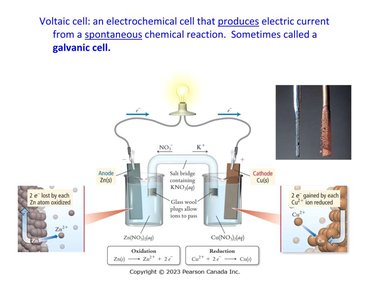
A voltaic cell (also called a galvanic cell) is an electrochemical cell that produces electric current from a spontaneous chemical reaction. It consists of two half-cells connected by a salt bridge or porous disk, each containing an electrode and an electrolyte solution.
Anode: Electrode where oxidation occurs (loss of electrons).
Cathode: Electrode where reduction occurs (gain of electrons).
Electrons flow from the anode to the cathode through an external circuit.
The anode is labeled negative (source of electrons), and the cathode is labeled positive.
Example: In a cell with zinc and copper electrodes, zinc is oxidized at the anode, and copper ions are reduced at the cathode.
Chemical Reactions in Voltaic Cells
The overall cell reaction is the sum of the oxidation and reduction half-reactions. For example:
Zn(s) + Cu2+(aq) → Zn2+(aq) + Cu(s)
When zinc metal is placed in a copper(II) sulfate solution, zinc atoms lose electrons (are oxidized), and copper ions gain electrons (are reduced), resulting in the deposition of copper metal.
Features of Voltaic Cells
Voltaic cells operate via spontaneous processes.
Electrodes are conductive surfaces for electron transfer.
Electrons flow from anode (oxidation) to cathode (reduction).
Mnemonic: Anode → "A negative electrode" (anode is negative in voltaic cells).
Maintaining Electrical Neutrality
As the cell operates, charge imbalances develop in each compartment:
Anode compartment accumulates cations (positive charge).
Cathode compartment accumulates anions (negative charge).
Neutrality is maintained by a salt bridge or porous glass disc that allows ion flow.
Salt bridge: permits cations to flow into the cathode compartment and anions into the anode compartment.

Cell Notation
Cell notation is a shorthand for representing voltaic cells:
Coefficients are omitted.
Phases are separated by commas; different phases by a vertical line.
Inert electrodes (e.g., Pt) are used if no solid is present.
Example: Zn(s) | Zn2+(aq) || Cu2+(aq) | Cu(s)
Cell Electromotive Force (EMF)
Definition and Origin
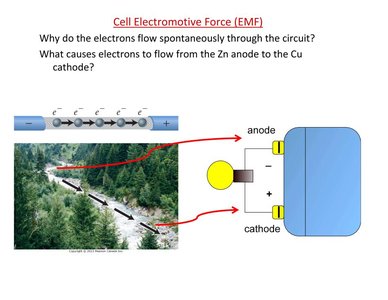
The cell electromotive force (EMF) is the potential difference between the two electrodes of a voltaic cell. It acts as the driving force for electron flow from the anode to the cathode.
Electrons flow spontaneously from higher to lower electrical potential energy.
The anode has higher potential energy than the cathode.
Features of EMF
Measured in volts (V), where 1 V = 1 J C-1.
Denoted as or .
Also called cell potential or cell voltage.
Always positive for a voltaic cell.
Units in Electrochemistry:
Coulomb (C): Unit of electric charge.
Ampere (A): Unit of current (1 A = 1 C s-1).
Volt (V): Unit of potential difference (1 V = 1 J C-1).
Faraday constant (F): Charge of one mole of electrons (1 F = 96,485 C mol-1).
Standard Electrode Potentials
Definition and Use
Standard electrode potentials () are tabulated values for the reduction of species under standard conditions (1 M, 1 atm, 25°C). They allow calculation of the cell potential for any voltaic cell reaction.
The cell potential is the difference between the cathode and anode potentials:
Standard electrode potentials are intensive properties (do not change with reaction coefficients).
Example:
Half-Reaction | (V) |
|---|---|
Cu2+(aq) + 2e- → Cu(s) | 0.34 |
F2(g) + 2e- → 2F-(aq) | 2.87 |

The Standard Hydrogen Electrode (SHE)
The standard hydrogen electrode (SHE) is the reference electrode with an assigned potential of 0 V. It consists of a platinum electrode in 1 M H+ solution with H2 gas at 1 bar.
All other electrode potentials are measured relative to the SHE.

Notes on Standard Electrode Potentials
The more positive the , the greater the driving force for reduction.
In a voltaic cell, the cathode is the half-cell with the most positive .
The greater the difference in values, the greater the cell potential.

Oxidizing and Reducing Agents
Identifying Strengths
The values of can be used to gauge the strength of oxidizing and reducing agents:
The more positive the , the stronger the oxidizing agent (left side of the table).
The more negative the , the stronger the reducing agent (right side of the table).
Example: Fe(s) is a stronger reducing agent than Mn2+(aq) or I-(aq) because it has a more negative value.

Practice Problems and Applications
Calculating Cell Potentials
To calculate the cell potential under standard conditions:
Identify the half-reactions and their standard reduction potentials.
Use the formula:
Example: For the reaction Mg(s) + Cu2+(aq) → Cu(s) + Mg2+(aq):
Cu2+(aq) + 2e- → Cu(s) V
Mg2+(aq) + 2e- → Mg(s) V
V
Identifying the Best Reducing Agent
The best reducing agent is the species with the most negative standard reduction potential.
For example, among Ni2+, Zn2+, Ag+, Ag, and Zn, Zn (with V) is the best reducing agent.
Summary Table: Selected Standard Electrode Potentials
Half-Reaction | (V) |
|---|---|
F2(g) + 2e- → 2F-(aq) | 2.87 |
MnO4-(aq) + 8H+(aq) + 5e- → Mn2+(aq) + 4H2O(l) | 1.51 |
Cu2+(aq) + 2e- → Cu(s) | 0.34 |
2H+(aq) + 2e- → H2(g) | 0.00 |
Zn2+(aq) + 2e- → Zn(s) | -0.76 |
Li+(aq) + e- → Li(s) | -3.05 |
Additional info: For a complete list, refer to a standard reduction potential table in your textbook or course materials.
