 Back
BackElectronic Structure of Atoms and Periodicity: Chapter 5 Study Notes
Study Guide - Smart Notes

Electronic Structure of Atoms
Electromagnetic Waves: Wavelength, Frequency, and Amplitude
Electromagnetic waves are characterized by three main properties: wavelength (λ), frequency (ν), and amplitude. Wavelength is the distance between two consecutive peaks, frequency is the number of cycles per second, and amplitude is the height of the wave from its central axis.
Wavelength (λ): Measured in meters (m), nanometers (nm), or angstroms (Å).
Frequency (ν): Measured in Hertz (Hz), or s-1.
Amplitude: Relates to the intensity of the wave.
Converting Between Wavelength and Frequency
The speed of light (c) relates wavelength and frequency:
Formula:
Where m/s
To find frequency:
To find wavelength:
Energy of Electromagnetic Radiation
The energy of a photon is directly proportional to its frequency and inversely proportional to its wavelength.
Formula:
Where J·s (Planck's constant)
Energy can be expressed as J/photon or kJ/mol (using Avogadro's number).
The Photoelectric Effect
The photoelectric effect occurs when light of sufficient frequency strikes a metal surface, causing the emission of electrons. This phenomenon supports the particle-like nature of light, as only photons above a threshold frequency can eject electrons, regardless of intensity.
Key Point: Demonstrates quantization of energy in light.
Example: UV light can cause photoemission from zinc, but visible light cannot.
Continuous vs. Line Spectra
A continuous spectrum contains all wavelengths within a range, while a line spectrum consists of discrete wavelengths emitted by atoms.
Continuous Spectrum: Produced by incandescent solids.
Line Spectrum: Produced by excited atoms (e.g., hydrogen emission lines).
Electron Transitions in the Bohr Model
Electron transitions between energy levels in the Bohr model result in emission or absorption of photons. The energy difference determines the wavelength and frequency of the emitted or absorbed light.
Higher energy transitions: Shorter wavelength, higher frequency.
Lower energy transitions: Longer wavelength, lower frequency.
de Broglie Equation: Wavelength of Moving Objects
All matter exhibits wave-like properties. The de Broglie equation relates the wavelength of a moving object to its momentum.
Formula:
Where is mass and is velocity.
Example: Electrons have measurable wavelengths; macroscopic objects do not.
Quantum Numbers and Atomic Orbitals
Quantum Numbers: Principal (n), Angular Momentum (l), Magnetic (ml), Spin (ms)
Quantum numbers describe the properties of electrons in atoms:
Principal quantum number (n): Energy level (shell), n = 1, 2, 3, ...
Angular momentum quantum number (l): Subshell shape, l = 0 (s), 1 (p), 2 (d), 3 (f)
Magnetic quantum number (ml): Orientation, ml = -l to +l
Spin quantum number (ms): Electron spin, ms = +1/2 or -1/2
Writing Sets of Quantum Numbers
Each electron in an atom is described by a unique set of four quantum numbers. For example, an electron in a 2p orbital may have n=2, l=1, ml=0, ms=+1/2.
Example: 1s electron: n=1, l=0, ml=0, ms=+1/2 or -1/2
Identifying s and p Orbitals by Shape and Quantum Numbers
s orbitals are spherical, p orbitals are dumbbell-shaped. Their quantum numbers reflect these shapes.
s orbital: l=0, ml=0
p orbital: l=1, ml=-1, 0, or +1
Assigning Quantum Numbers to Electrons
For each electron, assign n, l, ml, and ms based on its position in the atom.
Example: 3d electron: n=3, l=2, ml=-2 to +2, ms=+1/2 or -1/2
Electron Configuration and Periodicity
Electron Shielding and Subshell Energy Order
Electron shielding occurs when inner electrons reduce the effective nuclear charge felt by outer electrons, affecting subshell energy order.
Order of subshells: 1s < 2s < 2p < 3s < 3p < 4s < 3d < 4p < 5s < 4d < 5p < 6s < 4f < 5d < 6p < 7s < 5f < 6d < 7p
Additional info: Subshells fill in order of increasing energy, not strictly by n.
Predicting Subshell Filling Order
The periodic table helps predict the order of subshell filling. Electrons fill the lowest available energy subshell first (Aufbau principle).
Example: 4s fills before 3d.
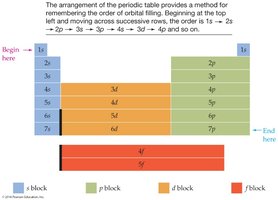
Assigning Electron Configurations
Electron configurations are written using subshell notation and noble gas core abbreviations.
Example: Sodium: [Ne] 3s1
Ground state: Lowest energy configuration.
Orbital Filling Diagrams and Unpaired Electrons
Orbital filling diagrams use arrows to represent electron spins in subshells. The number of unpaired electrons can be determined from these diagrams.
Example: Oxygen: 1s2 2s2 2p4 (two unpaired electrons in 2p)
Periodic Trends: Atomic Radii
Periodic Trend in Atomic Radii
Atomic radius decreases across a period (left to right) due to increased nuclear charge, and increases down a group due to added electron shells.
Across a period: Decreases
Down a group: Increases
Predicting Relative Size of Atoms
The position of an element on the periodic table allows prediction of its atomic size.
Example: Lithium is larger than fluorine; sodium is larger than lithium.
