 Back
BackElectronic Structure of Atoms and Quantum Mechanics
Study Guide - Smart Notes

Electronic Structure of Atoms
Introduction to Electronic Structure
The electronic structure of atoms is fundamental to understanding chemical properties and reactivity. This topic explores how electrons are arranged in atoms and the quantum mechanical principles that govern their behavior.
Waves and Electromagnetic Radiation
Wave Properties
Electromagnetic radiation exhibits wave-like behavior, characterized by wavelength and frequency.
Wavelength (λ): The distance between corresponding points on adjacent waves, such as from peak to peak or trough to trough.
Frequency (ν): The number of waves passing a given point per unit time. For waves traveling at the same velocity, a longer wavelength means a lower frequency.
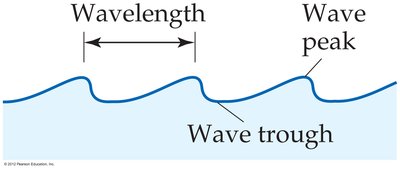
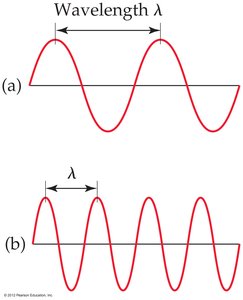
Electromagnetic Spectrum
All electromagnetic radiation travels at the speed of light (c), which is m/s. The relationship between wavelength and frequency is given by:
The electromagnetic spectrum includes gamma rays, X-rays, ultraviolet, visible light, infrared, microwaves, and radio waves. The visible region is a small part of this spectrum.
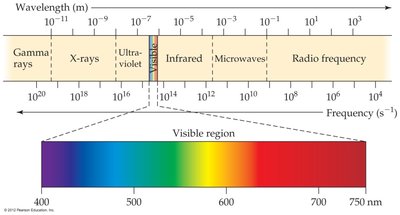
The Nature of Energy
Quantization of Energy
The wave nature of light alone cannot explain phenomena such as objects glowing when heated. Max Planck proposed that energy is quantized and comes in discrete packets called quanta.

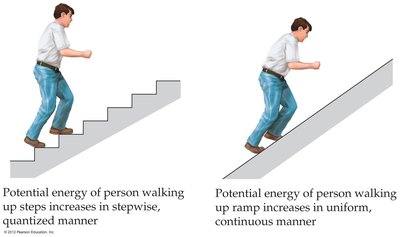
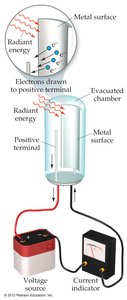
The Photoelectric Effect
Albert Einstein used Planck's idea to explain the photoelectric effect, showing that energy is proportional to frequency:
where is Planck's constant ( J·s).
Energy and Wavelength
If the wavelength of light is known, the energy of a photon can be calculated using:
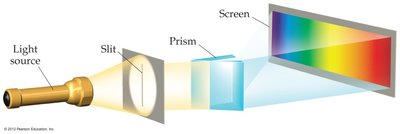
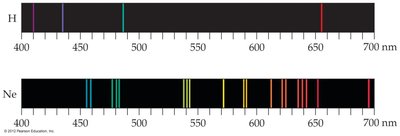
Atomic Emission Spectra
Atoms and molecules emit energy in discrete wavelengths, producing a line spectrum rather than a continuous spectrum. This observation was key to the development of quantum theory.
Bohr Model of the Atom
Bohr's Postulates
Niels Bohr explained atomic spectra by proposing:
Electrons occupy only certain allowed orbits (energy levels).
Electrons in permitted orbits have specific energies and do not radiate energy while in those orbits.
Energy is absorbed or emitted only when an electron moves between allowed energy states, with the energy change given by .
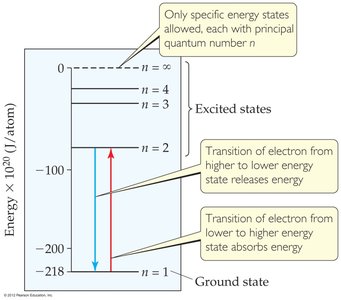
Energy Transitions
The energy absorbed or emitted during electron transitions is calculated by:
where is the Rydberg constant ( m), and and are the initial and final energy levels.
The Wave Nature of Matter
de Broglie Hypothesis
Louis de Broglie proposed that matter, like light, exhibits wave properties. The wavelength associated with a particle is:
where is mass and is velocity.
The Uncertainty Principle
Heisenberg's Principle
Werner Heisenberg showed that it is impossible to know both the position and momentum of a particle with absolute precision:
This uncertainty is significant for electrons in atoms.
Quantum Mechanics and Atomic Orbitals
Schrödinger Equation
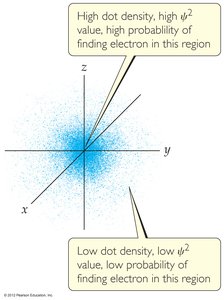
Erwin Schrödinger developed quantum mechanics, a mathematical framework that incorporates both wave and particle nature of matter. The wave function () describes the probability of finding an electron in a particular region.
The probability density is given by .
Quantum Numbers
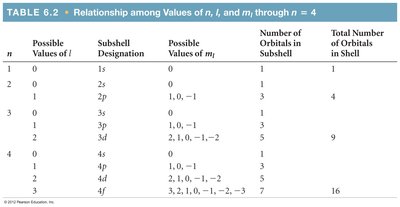
Solving the Schrödinger equation yields orbitals, each described by a set of quantum numbers:
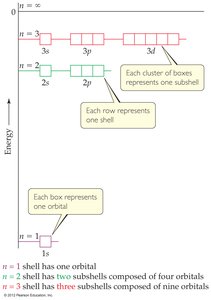
Principal quantum number (n): Indicates energy level (n = 1, 2, 3, ...).
Angular momentum quantum number (l): Defines orbital shape (l = 0 to n-1).
Magnetic quantum number (m_l): Describes orbital orientation (m_l = -l to +l).
Spin quantum number (m_s): Describes electron spin (+1/2 or -1/2).
Types of Orbitals
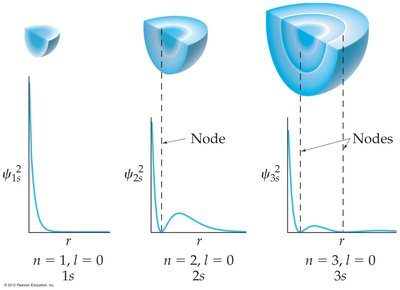
s orbitals (l = 0): Spherical shape, increasing radius with n.
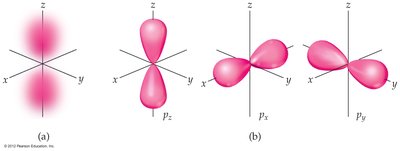
p orbitals (l = 1): Two lobes with a node between them.
d orbitals (l = 2): Four have four lobes; one resembles a p orbital with a doughnut shape.
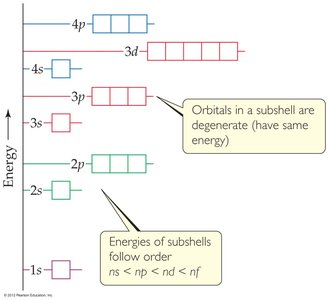
Energy Levels and Degeneracy
In hydrogen, orbitals with the same n have the same energy (degenerate). In multi-electron atoms, electron-electron repulsion removes this degeneracy.
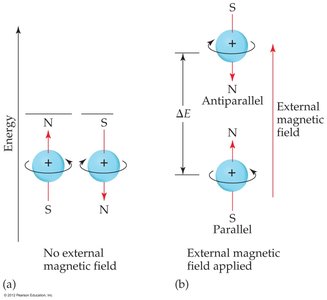
Electron Spin and the Pauli Exclusion Principle
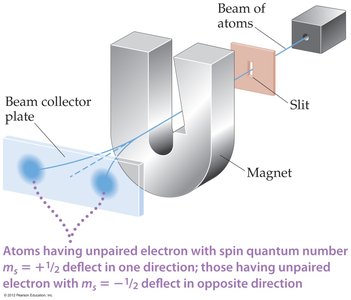
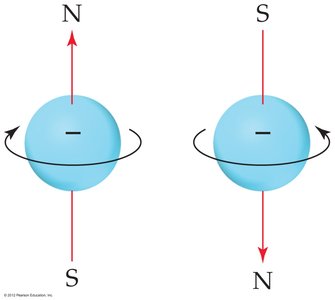
Spin Quantum Number (ms)
Electrons have an intrinsic property called spin, which can be +1/2 or -1/2. No two electrons in the same atom can have the same set of quantum numbers (Pauli Exclusion Principle).
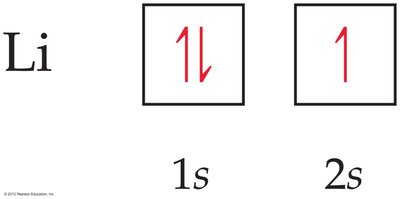
Electron Configurations and Orbital Diagrams
Electron Configurations
The electron configuration of an atom shows the distribution of electrons among orbitals. Each component consists of:
A number (energy level)
A letter (type of orbital)
A superscript (number of electrons in those orbitals)
Example: 4p5
Orbital Diagrams
Orbital diagrams use boxes to represent orbitals and arrows for electrons. The direction of the arrow indicates the electron's spin.
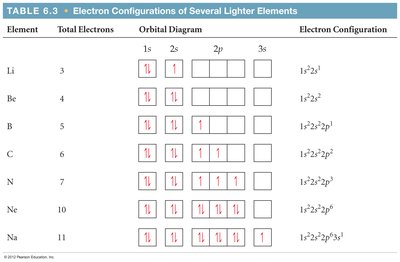
Hund's Rule
For degenerate orbitals (same energy), the lowest energy arrangement maximizes the number of electrons with the same spin.
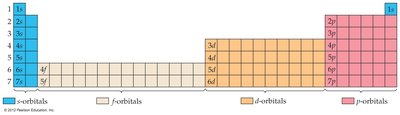
The Periodic Table and Electron Configurations
Filling Order and Blocks
Orbitals are filled in order of increasing energy. The periodic table is divided into blocks (s, p, d, f) corresponding to the type of orbital being filled.
Electron Configuration Anomalies
Some elements have electron configurations that differ from the expected order due to the close energy of s and d (or f) orbitals. For example, chromium is [Ar] 4s1 3d5 instead of [Ar] 4s2 3d4.
Additional info: These anomalies also occur in f-block elements, where electron stability is enhanced by half-filled or fully filled subshells.
