 Back
BackElectronic Structure of Atoms (Part 2): The Bohr Model, Quantum Mechanics, and Atomic Orbitals
Study Guide - Smart Notes

Electronic Structure of Atoms
The Bohr Model of the Atom
The Bohr model, developed by Niels Bohr in 1912-1913, was a major advancement in understanding atomic structure, particularly for the hydrogen atom. It introduced the concept of quantized energy levels for electrons.
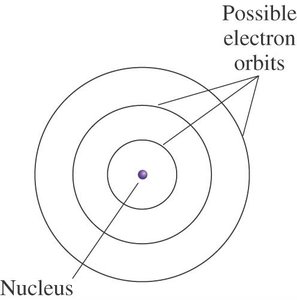
Electrons exist in specific regions (orbits) at various distances from the nucleus. These orbits are analogous to planets revolving around the sun.
Quantization of Energy: Electrons can only occupy certain allowed energy levels and cannot exist between them.
Ground State and Excited States: The lowest energy state is called the ground state. Any higher energy state is an excited state.
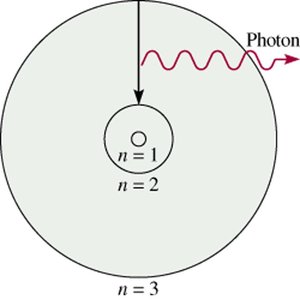
Energy Transitions: When an electron moves from a higher to a lower energy level, it emits a photon of light. Conversely, absorption of energy can promote an electron to a higher energy level.
Key Points:
Only certain orbits with specific radii and energies are permitted for electrons in a hydrogen atom.
Energy is emitted or absorbed as a photon when an electron transitions between energy levels.
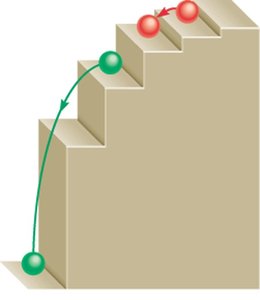
Quantization Analogy: The quantized nature of energy levels can be visualized as steps on a staircase—electrons can only occupy the steps, not the spaces in between.
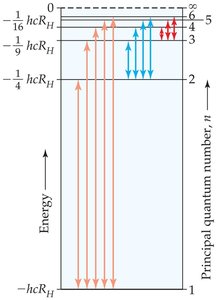
Energy Calculations in the Bohr Model
The energy difference between two levels determines the energy of the photon emitted or absorbed during a transition. The energy levels for the hydrogen atom are given by:
where is the Rydberg constant ( J), and is the principal quantum number (1, 2, 3, ...).
The energy change for a transition is:
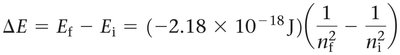
Where and are the initial and final energy levels, respectively.
Positive : Energy is absorbed (electron moves to a higher level).
Negative : Energy is released (electron falls to a lower level).
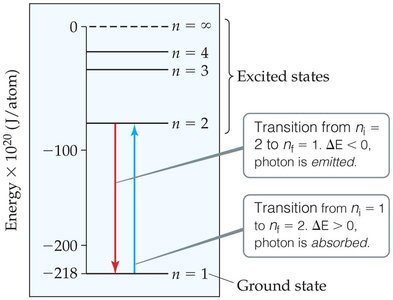
Example Calculation: To find the wavelength () of a photon emitted when an electron drops from to :
Calculate using the formula above.
Relate energy to wavelength:
Solve for :
where is Planck's constant ( J·s) and is the speed of light ( m/s).
Limitations of the Bohr Model
Accurately predicts spectral lines for hydrogen but fails for atoms with more than one electron.
Does not explain why electron energies are quantized or why electrons are restricted to certain orbits.
The Dual Nature of the Electron
Louis de Broglie proposed that particles such as electrons have wave-like properties, similar to how light can behave as both a wave and a particle.
The wavelength () of a particle is given by:
where is mass, is velocity, and is Planck's constant.
Wave properties are significant only for submicroscopic objects due to the small value of .
The Heisenberg Uncertainty Principle
Werner Heisenberg showed that it is impossible to know both the position and momentum of a particle with absolute precision. This is known as the uncertainty principle:
This principle is fundamental to quantum mechanics and limits our ability to describe the exact behavior of electrons in atoms.
Quantum Mechanics and Atomic Orbitals
Erwin Schrödinger developed quantum mechanics, a mathematical framework that incorporates both the wave and particle nature of matter. The solutions to Schrödinger's equation are called wave functions (), and the square of the wave function () gives the probability density of finding an electron in a particular region of space.
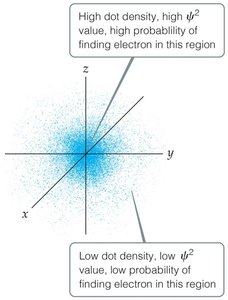
Key Points:
Each wave function corresponds to an orbital, a region in space where the probability of finding an electron is high.
Orbitals are described by a set of three quantum numbers: , , and .
Quantum Numbers
Principal Quantum Number (): Indicates the energy level and size of the orbital. Values:
Angular Momentum Quantum Number (): Defines the shape of the orbital. Values: to
Magnetic Quantum Number (): Describes the orientation of the orbital in space. Values: to
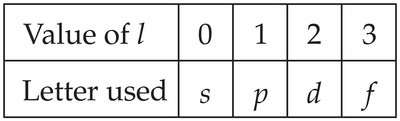
The letters s, p, d, f are used to designate the different values of :
Value of l | 0 | 1 | 2 | 3 |
|---|---|---|---|---|
Letter used | s | p | d | f |
The relationship among , , and determines the number and types of orbitals in each energy level:
n | Possible Values of l | Subshell Designation | Possible Values of ml | Number of Orbitals in Subshell | Total Number of Orbitals in Shell |
|---|---|---|---|---|---|
1 | 0 | 1s | 0 | 1 | 1 |
2 | 0 | 2s | 0 | 1 | 4 |
2 | 1 | 2p | -1, 0, 1 | 3 | |
3 | 0 | 3s | 0 | 1 | 9 |
3 | 1 | 3p | -1, 0, 1 | 3 | |
3 | 2 | 3d | -2, -1, 0, 1, 2 | 5 | |
4 | 0 | 4s | 0 | 1 | 16 |
4 | 1 | 4p | -1, 0, 1 | 3 | |
4 | 2 | 4d | -2, -1, 0, 1, 2 | 5 | |
4 | 3 | 4f | -3, -2, -1, 0, 1, 2, 3 | 7 |
Shapes of Atomic Orbitals
s Orbitals
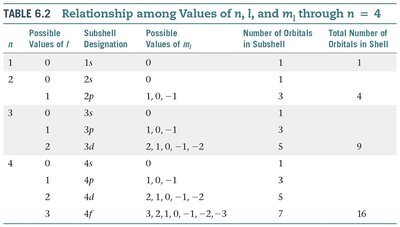
s orbitals () are spherical in shape. The size of the sphere increases with increasing (e.g., 1s, 2s, 3s).
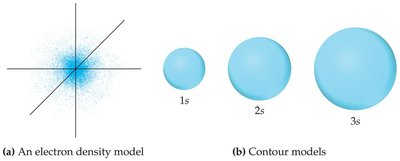
p Orbitals
p orbitals () have two lobes with a node (region of zero probability) between them. There are three p orbitals (px, py, pz) in each energy level where .
d and f Orbitals
d orbitals () have more complex shapes, typically four lobes, with one resembling a p orbital with a doughnut around the center. There are five d orbitals in each energy level where .
f orbitals () have even more complex shapes and there are seven f orbitals in each energy level where .
Summary Table: Quantum Numbers and Orbitals
Quantum Number | Symbol | Meaning | Possible Values |
|---|---|---|---|
Principal | n | Energy level, size of orbital | 1, 2, 3, ... |
Angular Momentum | l | Shape of orbital | 0 to n-1 |
Magnetic | ml | Orientation of orbital | -l to +l |
Additional info: The quantum mechanical model replaces the concept of fixed orbits with orbitals, which are regions of space with a high probability of finding an electron. This model successfully explains the structure and behavior of all atoms, not just hydrogen.
