 Back
BackElectronic Structure of Atoms: Waves, Electromagnetic Radiation, and Atomic Spectra
Study Guide - Smart Notes

Electronic Structure of Atoms
Waves and Electromagnetic Radiation
Understanding the electronic structure of atoms requires a foundational knowledge of electromagnetic radiation, which moves as waves through space at the speed of light. These waves possess distinct properties that are crucial for describing atomic behavior.
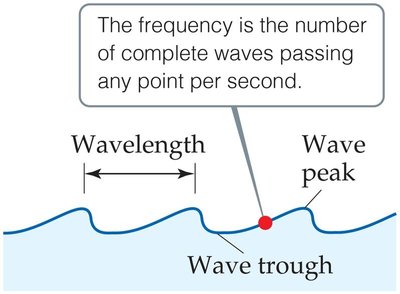
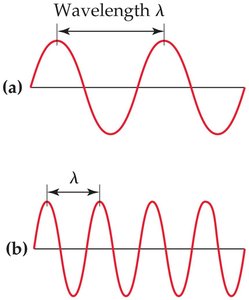
Wavelength (\(\lambda\)): The distance between corresponding points on adjacent waves, such as from peak to peak or trough to trough.
Amplitude: The vertical distance from the midline of a wave to its peak or trough, representing the wave's energy.
Frequency (\(\nu\)): The number of complete waves passing a given point per second, measured in Hertz (Hz).
Relationship: For waves traveling at the same velocity, a longer wavelength corresponds to a lower frequency, and vice versa.
Equation: The speed of light (\(c\)) relates wavelength and frequency:
Where:
\(c\) = speed of light (3.00 × 108 m/s)
\(\lambda\) = wavelength (m)
\(\nu\) = frequency (s−1 or Hz)
Nature of Electromagnetic Waves
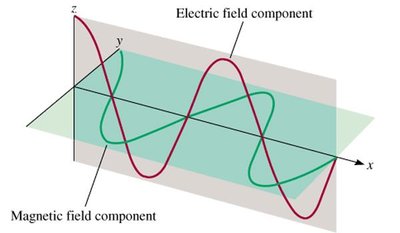
Electromagnetic radiation consists of oscillating electric and magnetic fields perpendicular to each other and to the direction of wave propagation. Maxwell (1873) proposed that visible light is a form of electromagnetic waves.
Types of Electromagnetic Radiation
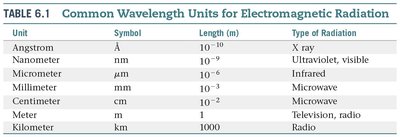
Electromagnetic radiation encompasses a wide range of wavelengths and energies, from gamma rays to radio waves. Each type is characterized by its wavelength and frequency, and different units are used to describe these ranges.
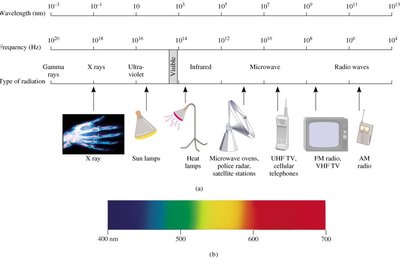
Unit | Symbol | Length (m) | Type of Radiation |
|---|---|---|---|
Angstrom | Å | 10−10 | X ray |
Nanometer | nm | 10−9 | Ultraviolet, visible |
Micrometer | μm | 10−6 | Infrared |
Millimeter | mm | 10−3 | Microwave |
Centimeter | cm | 10−2 | Microwave |
Meter | m | 1 | Television, radio |
Kilometer | km | 1000 | Radio |
Calculating Wavelength and Frequency
To convert frequency to wavelength:
Example: A photon with frequency 6.0 × 104 Hz has a wavelength:
Electronic Properties Not Explained by Waves
Limitations of Classical Wave Theory
Three phenomena could not be explained by classical wave theory:
Blackbody Radiation: The emission of light from hot objects.
Photoelectric Effect: The emission of electrons from metal surfaces when exposed to light.
Emission Spectra: The emission of light from electronically excited gas atoms.
Blackbody Radiation and Quantum Theory
The wave nature of light fails to explain why objects glow when heated. Max Planck proposed that energy is emitted in discrete packets called quanta (singular: quantum), revolutionizing physics.

Planck's Quantum Theory: Atoms and molecules emit energy only in certain discrete quantities.
Quantum: The smallest quantity of energy that can be emitted or absorbed as electromagnetic radiation.
Energy of a quantum:
Where h is Planck’s constant (6.63 × 10−34 J·s).
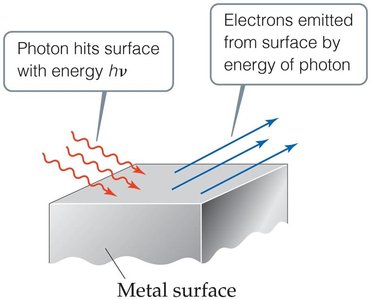
The Photoelectric Effect
Einstein explained the photoelectric effect by proposing that light consists of particles called photons. Each photon carries energy proportional to its frequency.
Electrons are ejected from metal surfaces only when light of sufficient frequency (threshold frequency) strikes the surface.
Energy is proportional to frequency:
Example: Calculate the energy of X-ray photons with wavelength 0.154 nm:
Atomic Emissions and Spectra
Emission Spectra of Atoms
When atoms and molecules emit energy, they produce emission spectra. Unlike a continuous spectrum (rainbow), atoms emit only specific wavelengths, resulting in a line spectrum. Each element has a unique line spectrum.

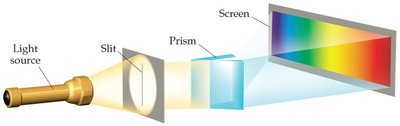
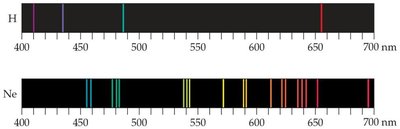
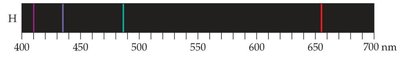
Continuous Spectrum: Produced by white light, contains all wavelengths.
Line Spectrum: Produced by excited atoms, contains only specific wavelengths.
Application: Line spectra are used to identify elements and study atomic structure.
Example: The hydrogen spectrum consists of discrete lines at specific wavelengths, revealing quantized energy levels in atoms.
Additional info: The study of atomic spectra led to the development of quantum mechanics and the understanding of electronic structure in atoms.
