 Back
BackElectrons in Atoms: Light, Quantum Theory, and Electron Configurations
Study Guide - Smart Notes

Electrons in Atoms
Introduction
The arrangement of electrons in atoms is fundamental to understanding chemical behavior. This chapter explores the wave and particle models of light, the quantization of energy, atomic emission spectra, and the development of quantum theory and electron configurations.
Light and Quantized Energy
Wave Nature of Light
Electromagnetic radiation is a form of energy that exhibits wavelike behavior as it travels through space. Visible light, microwaves, X-rays, and radio waves are all types of electromagnetic radiation. All waves can be described by their wavelength (λ), frequency (ν), amplitude, and speed.
Wavelength (λ): The shortest distance between equivalent points on a wave, usually measured in meters, centimeters, or nanometers.
Frequency (ν): The number of waves that pass a given point per second, measured in hertz (Hz).
Amplitude: The height of the wave from the origin to the crest or trough.
Speed of Light (c): All electromagnetic waves travel at m/s in a vacuum.
Relationship:
Wavelength and frequency are inversely related: as one increases, the other decreases.


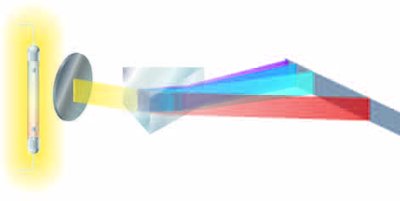
Electromagnetic Spectrum
The electromagnetic spectrum includes all forms of electromagnetic radiation, differing only in wavelength and frequency. Energy increases with increasing frequency.
Particle Nature of Light
While the wave model explains much of light's behavior, it cannot explain phenomena such as the emission of specific frequencies of light by heated objects or the photoelectric effect.
Quantum: The minimum amount of energy that can be gained or lost by an atom.
Planck's Constant (h): J·s
Energy of a quantum:
Photoelectric Effect: Electrons are emitted from a metal's surface when light of a certain frequency shines on it.
Photon: A particle of electromagnetic radiation with no mass that carries a quantum of energy.
Energy of a photon:


Atomic Emission Spectra
When atoms absorb energy, their electrons become excited and emit light as they return to lower energy states. The atomic emission spectrum of an element is the set of frequencies of electromagnetic waves emitted by its atoms. Each element's spectrum is unique and consists of discrete lines, not a continuous range.
Application: Flame tests and emission spectra are used to identify elements.


Quantum Theory and the Atom
Bohr Model of the Atom
Niels Bohr proposed that electrons move in specific, quantized orbits around the nucleus. Each orbit corresponds to a specific energy level, and transitions between levels result in the emission or absorption of photons.
Ground State: The lowest energy state of an atom.
Excited State: Higher energy states achieved when electrons absorb energy.
Energy Change:
Quantum Number (n) | Orbit Radius (nm) | Relative Energy |
|---|---|---|
1 | 0.0529 | |
2 | 0.212 | |
3 | 0.476 | |
4 | 0.846 | |
5 | 1.32 | |
6 | 1.90 | |
7 | 2.59 |
Quantum Mechanical Model of the Atom
Louis de Broglie proposed that electrons have wave-like properties. The quantum mechanical model, developed by Schrödinger, treats electrons as waves and predicts their probable locations in atomic orbitals.
de Broglie Equation:
Heisenberg Uncertainty Principle: It is impossible to know both the position and velocity of an electron simultaneously.
Atomic Orbital: A three-dimensional region around the nucleus where an electron is likely to be found.
Principal Quantum Number (n): Indicates the size and energy of the orbital.
Energy Sublevels: Each principal energy level contains sublevels labeled s, p, d, and f.




Electron Configurations
Ground-State Electron Configurations
The arrangement of electrons in an atom is called its electron configuration. Electrons fill orbitals according to three rules:
Aufbau Principle: Electrons occupy the lowest energy orbitals first.
Pauli Exclusion Principle: A maximum of two electrons may occupy a single orbital, and they must have opposite spins.
Hund's Rule: Single electrons with the same spin occupy each equal-energy orbital before additional electrons with opposite spins can occupy the same orbitals.

Orbital Diagrams and Electron Configuration Notation
Electron configurations can be represented using orbital diagrams (boxes and arrows) or electron configuration notation (numbers and letters with superscripts). Noble-gas notation is a shorthand method using the symbol of the previous noble gas in brackets.
Element | Atomic Number | Electron Configuration Notation |
|---|---|---|
Hydrogen | 1 | 1s1 |
Helium | 2 | 1s2 |
Lithium | 3 | 1s22s1 |
Beryllium | 4 | 1s22s2 |
Boron | 5 | 1s22s22p1 |
Carbon | 6 | 1s22s22p2 |
Nitrogen | 7 | 1s22s22p3 |
Oxygen | 8 | 1s22s22p4 |
Fluorine | 9 | 1s22s22p5 |
Neon | 10 | 1s22s22p6 |

Valence Electrons and Electron-Dot Structures
Valence electrons are electrons in the atom's outermost orbitals and determine chemical properties. Electron-dot structures visually represent valence electrons as dots around the element's symbol.
Element | Electron Configuration | Electron-Dot Structure |
|---|---|---|
Lithium | 1s22s1 | Li· |
Beryllium | 1s22s2 | Be·· |
Boron | 1s22s22p1 | B··· |
Carbon | 1s22s22p2 | C···· |
Nitrogen | 1s22s22p3 | N····· |
Oxygen | 1s22s22p4 | O······ |
Fluorine | 1s22s22p5 | F······· |
Neon | 1s22s22p6 | Ne········ |
Summary of Key Equations
EM Wave relationship:
Energy of a quantum:
Energy of a photon:
Energy change of an electron:
de Broglie’s equation:
Additional info:
Atomic emission spectra and electron configurations are essential for identifying elements and understanding their chemical properties.
Quantum theory explains the discrete nature of energy levels and the probabilistic location of electrons in atoms.
