 Back
BackElementary Chemical Kinetics: Principles, Rate Laws, and Applications
Study Guide - Smart Notes

Elementary Chemical Kinetics
Introduction to Chemical Kinetics
Chemical kinetics is the study of the rates at which chemical reactions occur and the factors that influence these rates. Understanding kinetics provides insight into reaction mechanisms and is essential for predicting how fast a reaction will proceed under given conditions.
Reaction Rate: The change in concentration of a reactant or product per unit time.
Applications: Drug release, drug degradation, and isomerization in pharmaceutical sciences.
Reaction Mechanisms and Molecularity
A reaction mechanism describes the sequence of elementary steps that make up the overall reaction. Each step is characterized by its molecularity, which is the number of reactant molecules involved in that step.
Elementary Step: A single step in a reaction mechanism involving a specific molecularity (unimolecular, bimolecular, etc.).
Intermediate: A species produced in one step and consumed in another, not present in the overall reaction equation.
Example: The reaction NO2 (g) + CO (g) → NO (g) + CO2 (g) proceeds via two elementary steps, with NO3 as an intermediate.
Factors Affecting Reaction Rate
Concentration of reactants
Temperature and pressure
Solvent properties (polarity, ionic strength)
Surface area of solid reactants
Action of catalysts
Mathematical Expression of Reaction Rate
For a reaction A + B → C, the rate can be expressed as:
Rate Laws and Reaction Order
General Rate Law
The rate law relates the rate of reaction to the concentration of reactants, each raised to a power (the order with respect to that reactant):
k: Rate constant (depends on temperature and pressure)
α, β: Reaction orders with respect to A and B (not necessarily stoichiometric coefficients)
Overall order: α + β
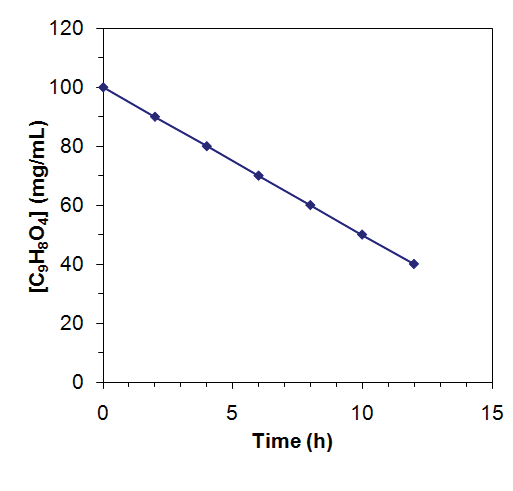
Zero-Order Reactions
In zero-order reactions, the rate is independent of the concentration of reactants.
Rate law:
Integrated rate law:
Units of k: concentration·time-1 (e.g., mol·L-1·s-1)
Graph: [A] vs. time is linear with slope -k
![Zero-order reaction: [C9H8O4] vs time plot](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/4fd76138_image_13.png)
First-Order Reactions
In first-order reactions, the rate depends linearly on the concentration of one reactant.
Rate law:
Integrated rate law: or
Units of k: time-1 (e.g., s-1)
Graph: ln[A] vs. time is linear with slope -k
![First-order reaction: [C16H21NO3] vs time plot](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/4fd76138_image_16.png)
Second-Order Reactions
Second-order reactions can depend on the concentration of one reactant squared or the product of two reactant concentrations.
Rate law (type I):
Integrated rate law (type I):
Rate law (type II):
Units of k: concentration-1·time-1 (e.g., L·mol-1·s-1)
Graph: 1/[A] vs. time is linear with slope k
![Second-order reaction: 1/[HI] vs time plot](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/4fd76138_image_20.png)
Summary Table: Rate Laws and Plots
Order | Rate Law | Integrated Rate Law | Units of k | Linear Plot |
|---|---|---|---|---|
Zero | concentration·time-1 | [A] vs. time | ||
First | time-1 | ln[A] vs. time | ||
Second | or | concentration-1·time-1 | 1/[A] vs. time |
Half-Life and Shelf-Life
Definitions
Half-life (t1/2): Time required for the concentration of a reactant to decrease by half.
Shelf-life (t0.9): Time required for 10% decomposition of a pharmaceutical product (i.e., when 90% remains).
Formulas:
Zero-order: ,
First-order: ,
Second-order: ,
Reversible Reactions and Equilibrium
Some reactions are reversible, proceeding in both forward and backward directions until equilibrium is reached.
At equilibrium: Rate of forward reaction equals rate of backward reaction.
Equilibrium constant (K):
Temperature Dependence of Rate Constant: Arrhenius Equation
The Arrhenius equation describes how the rate constant (k) varies with temperature (T):
A: Pre-exponential (frequency) factor
Ea: Activation energy (J·mol-1)
R: Gas constant (8.314 J·K-1·mol-1)
T: Absolute temperature (K)
Logarithmic form:
A plot of ln k vs. 1/T yields a straight line with slope .
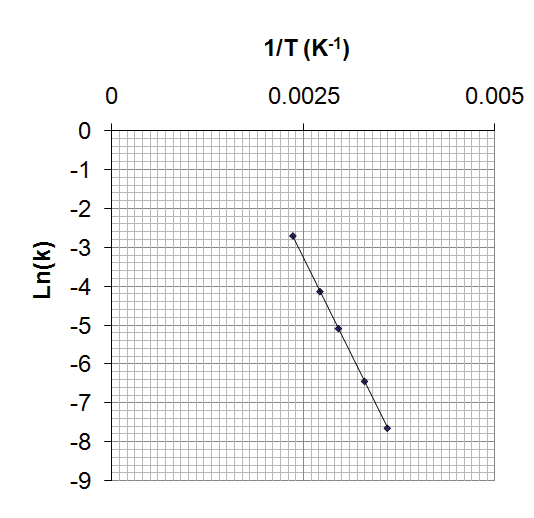
Two-Point Arrhenius Equation
To calculate activation energy or predict k at a new temperature using two data points:
Applications in Pharmacy
Drug Release: Kinetics governs how drugs are released from dosage forms (e.g., tablets, capsules).
Drug Degradation: Understanding kinetics helps predict shelf-life and stability of pharmaceuticals.
Enzyme Kinetics: Important for understanding drug metabolism and action in the body.
Appendix: The Gas Constant in Various Units
Value | Units |
|---|---|
8.31451 | J·K-1·mol-1 |
8.20578 × 10-2 | L·atm·K-1·mol-1 |
8.31451 × 10-2 | L·bar·K-1·mol-1 |
8.31451 | Pa·m3·K-1·mol-1 |
62.364 | L·Torr·K-1·mol-1 |
1.98722 | cal·K-1·mol-1 |
Additional info: The study of chemical kinetics is essential for understanding reaction mechanisms, optimizing pharmaceutical formulations, and ensuring drug safety and efficacy through proper shelf-life determination.
