 Back
BackEnergy and Physical Properties: Intermolecular Forces, Solutions, and Concentration
Study Guide - Smart Notes

Energy and Physical Properties
The Three States of Matter
Matter exists in three primary states: solid, liquid, and gas. Each state is defined by the arrangement and energy of its particles, which are influenced by intermolecular forces.
Solids: Particles are closely packed in a fixed structure; strong intermolecular forces.
Liquids: Particles are close but can move past each other; moderate intermolecular forces.
Gases: Particles are far apart and move freely; weak intermolecular forces.
Intermolecular Forces
Types of Intermolecular Forces
Intermolecular forces are the attractions between molecules, ions, or atoms that determine the physical properties of substances. The main types are:
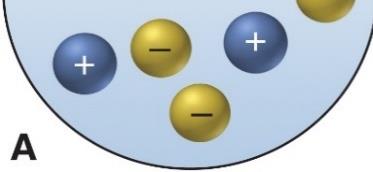
Ion-Ion Forces: Attraction between oppositely charged ions. Strongest type of intermolecular force.
Ion-Dipole Forces: Attraction between an ion and a polar molecule.
Dipole-Dipole Forces: Attraction between polar molecules.
Hydrogen Bonding: Special dipole-dipole interaction involving H bonded to N, O, or F.
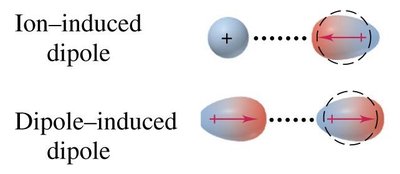
Ion-Induced Dipole Forces: Attraction between an ion and a nonpolar molecule, where the ion induces a dipole in the nonpolar molecule.
Dipole-Induced Dipole Forces: Attraction between a polar molecule and a nonpolar molecule, where the polar molecule induces a dipole in the nonpolar molecule.
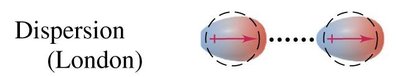
Dispersion (London) Forces: Weak attractions due to temporary dipoles in all molecules, especially significant in nonpolar molecules.

Ion-Ion and Ion-Induced Dipole Forces
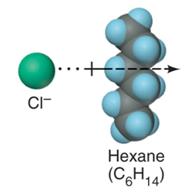
Ion-ion forces are the strongest, occurring between fully charged ions. Ion-induced dipole forces occur when an ion distorts the electron cloud of a nearby nonpolar molecule, inducing a temporary dipole.
Dispersion (London) Forces
Dispersion forces arise from temporary fluctuations in electron distribution, creating instantaneous dipoles that induce dipoles in neighboring particles. These forces are present in all molecules but are the only forces in nonpolar substances. Larger molecules have stronger dispersion forces, leading to higher melting and boiling points.
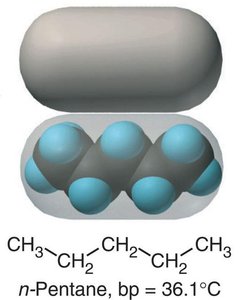
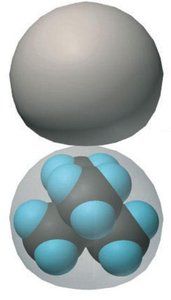
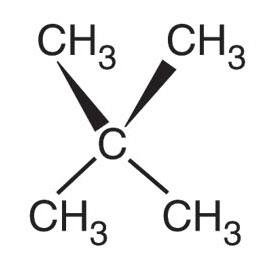
Molecular Shape and Intermolecular Contact
The shape of a molecule affects how much surface area is available for intermolecular contact, influencing the strength of dispersion forces and thus boiling points.
Linear molecules (e.g., n-pentane) have more contact points and stronger dispersion forces.
Compact molecules (e.g., neopentane) have fewer contact points and weaker dispersion forces.
Summary Table of Intermolecular Forces
The following table summarizes the main types of intermolecular forces, their models, basis of attraction, relative energies, and examples:
Force | Model | Basis of Attraction | Energy (kJ/mol) | Example |
|---|---|---|---|---|
Ion-dipole |
| Ion charge–dipole charge | 40–600 | Na+ in H2O |
H bond | H bonded to N, O, or F | Polar bond to H–dipole charge | 10–40 | H2O, HF |
Dipole-dipole | Dipole charges | Dipole charges | 5–25 | HCl, CH3Cl |
Ion-induced dipole |
| Ion charge–polarizable e- cloud | 3–15 | Fe2+ and O2 |
Dipole-induced dipole |
| Dipole charge–polarizable e- cloud | 2–10 | HCl and Cl2 |
Dispersion (London) |
| Polarizable e- clouds | 0.05–40 | F2, I2 |

Solutions and the Dissolving Process
Solutions, Suspensions, and Colloids
A solution is a homogeneous mixture of two or more substances in a single phase. The solvent is the major component, and the solute is the minor component. Water is the most common solvent, and solutions with water as the solvent are called aqueous solutions.
Suspension: Heterogeneous mixture with visible particles that settle out (e.g., sand in water).
Colloid: Heterogeneous mixture with particles that do not settle and exhibit the Tyndall effect (scattering of light).
Light Scattering and the Tyndall Effect
The Tyndall effect is observed when light passes through a colloid and is scattered, making the light beam visible. This effect distinguishes colloids from true solutions.

Electrolytes and Nonelectrolytes
Conductivity in Aqueous Solutions
Electrolytes are substances that conduct electricity when dissolved in water because they dissociate into ions. Nonelectrolytes do not conduct electricity because they do not form ions in solution.
Strong electrolytes: Completely dissociate into ions (e.g., NaCl).
Weak electrolytes: Partially dissociate into ions.
Nonelectrolytes: Do not dissociate (e.g., sugar).



Concentration of Solutions
Measuring Concentration
Concentration expresses the amount of solute in a given amount of solution. Common units include:
Percent concentration (%): Mass, volume, or mass/volume percent.
Parts per million (ppm) and parts per billion (ppb): Used for very dilute solutions.
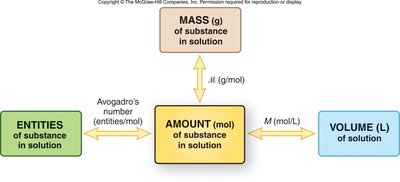
Molarity (M): Moles of solute per liter of solution.
Formulas:
Mass percent (w/w)%:
Volume percent (v/v)%:
Mass/volume percent (w/v)%:
ppm:
ppb:
Molarity:
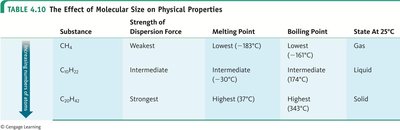
Sample Table: Effect of Molecular Size on Physical Properties
Substance | Strength of Dispersion Force | Melting Point | Boiling Point | State at 25°C |
|---|---|---|---|---|
CH4 | Weakest | Lowest (–183°C) | Lowest (–164°C) | Gas |
C5H12 | Intermediate | Intermediate (–130°C) | Intermediate (36°C) | Liquid |
C20H42 | Strongest | Highest (37°C) | Highest (343°C) | Solid |

Summary
Intermolecular forces determine the physical properties of substances and their states of matter.
Solutions, suspensions, and colloids differ in particle size and homogeneity.
Electrolytes conduct electricity in solution; nonelectrolytes do not.
Concentration can be measured in several ways, including percent, ppm, ppb, and molarity.
