 Back
BackEnergy and Physical Properties: Study Notes for General Chemistry
Study Guide - Smart Notes

Energy and Physical Properties
Energy
Energy is a fundamental concept in chemistry, representing the capacity to do work. Work involves moving something, and is mathematically defined as force multiplied by distance. There are several types of energy relevant to chemistry:
Potential Energy: Energy due to position or composition. Chemical energy is a form of potential energy stored within the structural units of chemical substances.
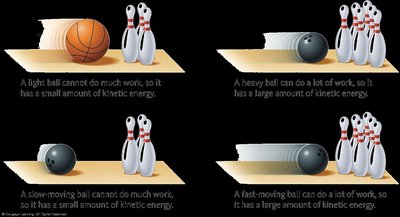
Kinetic Energy: Energy due to movement. The kinetic energy of an object is given by the equation: where m is mass and u is speed.
Thermal Energy: A type of kinetic energy associated with the random motion of atoms and molecules.
Heat: The transfer of thermal energy between two objects at different temperatures.


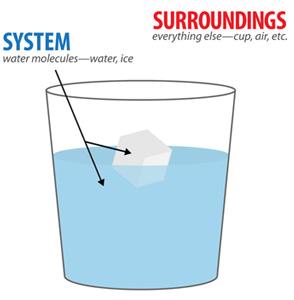
The System and Its Surroundings
In thermodynamics, the universe is divided into the system (the part of interest) and the surroundings (everything else). Systems can be:
Open: Exchange both matter and energy with surroundings.
Closed: Exchange energy but not matter.
Isolated: Exchange neither energy nor matter.
The Law of Energy Conservation
The First Law of Thermodynamics states that the total energy of the universe is constant. Energy is conserved and can be transferred as heat or work, but not created or destroyed.
Internal Energy (Esys): The sum of potential, chemical, and kinetic energies of all particles in the system.
Energy changes in the system are balanced by equal and opposite changes in the surroundings:
Exothermic and Endothermic Processes
Chemical reactions and physical changes can be classified based on heat flow:
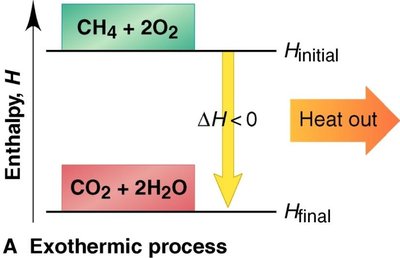
Exothermic: Releases heat; the enthalpy of the system decreases ().
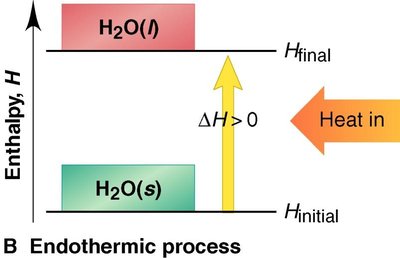
Endothermic: Absorbs heat; the enthalpy of the system increases ().
Units of Energy
Joule (J): SI unit of energy.
Calorie (cal): Energy needed to raise 1 g of water by 1°C.
British Thermal Unit (Btu):
Dietary Calorie (Cal):
Energy and Power
Power is the rate at which energy is produced or consumed, measured in watts (W):
Electric bills use kilowatt·hour as a unit of energy.
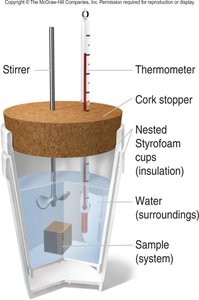
Calorimetry
Calorimetry is the measurement of heat produced or absorbed during a process. The specific heat capacity (c) is the amount of heat required to change the temperature of 1 gram of a substance by 1°C or 1 K.
Calorimeters are insulated devices used to measure energy transfer.
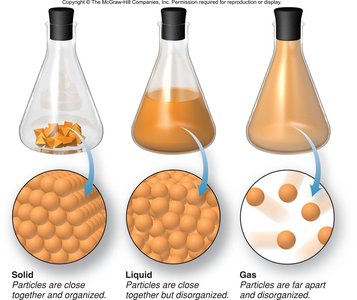
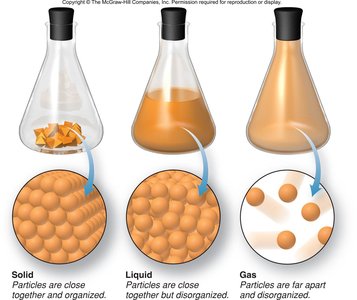
The Three States of Matter
Physical States
Matter exists in three primary states: solid, liquid, and gas. Each state has distinct physical properties:
Solids: Fixed shape and definite volume; particles are closely packed and organized.
Liquids: Indefinite shape but definite volume; particles are close together but disorganized.
Gases: Indefinite shape and volume; particles are far apart and disorganized.
Phase Changes
Phase changes involve the complete conversion of a sample from one physical state to another. Common phase changes include:
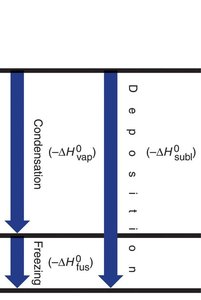
Melting: Solid to liquid (endothermic)
Freezing: Liquid to solid (exothermic)
Vaporizing/Boiling: Liquid to gas (endothermic)
Condensing: Gas to liquid (exothermic)
Sublimation: Solid to gas (endothermic)
Deposition: Gas to solid (exothermic)
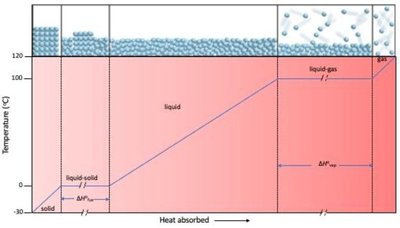
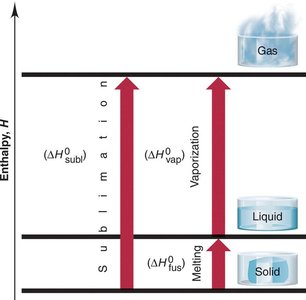
Enthalpy Changes in Phase Transitions
Heat of Fusion (): Energy required to melt 1 g of a solid at its melting point.
Heat of Vaporization (): Energy required to convert 1 g of liquid at its boiling point to gas.
Heat of Sublimation (): Energy required to sublime 1 g of solid at its sublimation temperature.
The Properties of Gases
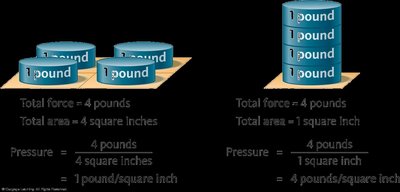
Gas Pressure
Pressure is the force exerted by gas molecules colliding with a surface, divided by the area of that surface:
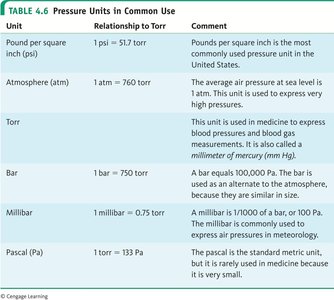
Pressure Units
Gas pressure is measured in several units. The most common are:
Unit | Relationship to Torr | Comment |
|---|---|---|
Pound per square inch (psi) | 1 psi = 51.7 torr | Common in the US |
Atmosphere (atm) | 1 atm = 760 torr | Average air pressure at sea level |
Torr | 1 torr = 1 mm Hg | Used in medicine and blood pressure |
Bar | 1 bar = 750 torr | Equals 100,000 Pa |
Millibar | 1 millibar = 0.75 torr | Used in meteorology |
Pascal (Pa) | 1 torr = 133 Pa | SI unit, rarely used in medicine |
The Gas Laws
The physical behavior of gases is described by four variables: pressure (P), temperature (T), volume (V), and amount (n, moles). The ideal gas law relates these variables:
R is the universal gas constant:
Pressure and Moles
At fixed temperature and volume, pressure is directly proportional to the amount of gas:
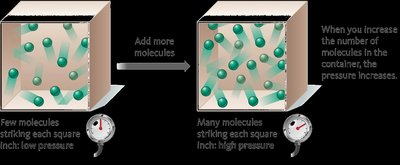
Pressure and Volume
At constant temperature, the volume occupied by a fixed amount of gas is inversely proportional to the external pressure:
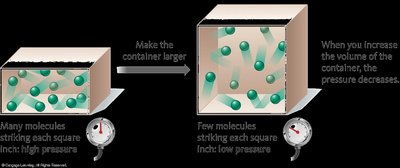
Pressure and Temperature
At fixed volume, pressure is directly proportional to temperature:
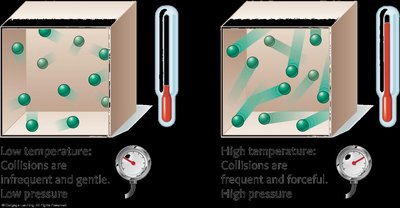
Standard Temperature and Pressure (STP)
STP: 1 atm, 0°C (273.15 K)
Standard molar volume: 22.4 L for 1 mol of ideal gas at STP
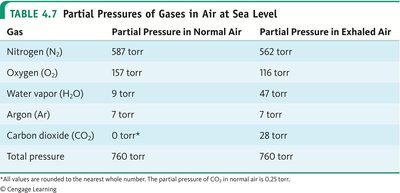
Gas Mixtures and Partial Pressures
In a gas mixture, each gas exerts a partial pressure. Dalton’s law states that the total pressure is the sum of the partial pressures:
Gas | Partial Pressure in Normal Air | Partial Pressure in Exhaled Air |
|---|---|---|
Nitrogen (N2) | 587 torr | 562 torr |
Oxygen (O2) | 157 torr | 116 torr |
Water vapor (H2O) | 9 torr | 47 torr |
Argon (Ar) | 7 torr | 7 torr |
Carbon dioxide (CO2) | 0 torr | 28 torr |
Total pressure | 760 torr | 760 torr |
Intermolecular Forces
Intramolecular vs Intermolecular Forces
Intramolecular forces (bonding forces) are found within a molecule and control its chemical properties. Intermolecular forces (nonbonding forces) are found between molecules and control physical properties such as melting point, boiling point, and density.
Types of Intermolecular Forces
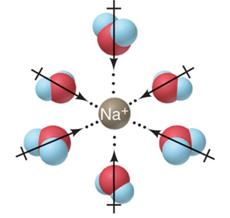
Ion-Ion: Forces between ions in solution; strongest intermolecular force.
Ion-Dipole: Forces between ions and polar molecules; second strongest.
Dipole-Dipole: Forces between two polar molecules; third strongest.
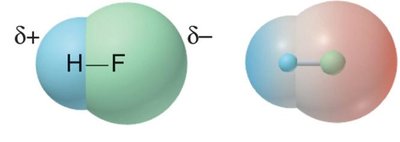
Hydrogen Bonding: Special type of dipole-dipole force; occurs when H is bonded to N, O, or F and interacts with lone pairs on N, O, or F in another molecule.

Hydrogen Bonding
Hydrogen bonding is the strongest dipole-dipole intermolecular force. It occurs when a hydrogen atom in a polar covalent bond (N-H, O-H, or F-H) is attracted to the lone pair of a highly electronegative atom (N, O, or F) in another molecule. Hydrogen bonding significantly affects physical properties such as boiling point and solubility.
Determining Hydrogen Bond Formation
Presence of N-H, O-H, or F-H polar covalent bonds
Presence of N, O, or F atoms with lone pairs
Both features must be provided by different molecules involved in H-bonding
Effects of Hydrogen Bonding
Hydrogen bonding changes physical properties drastically, such as increasing boiling points and affecting molecular structure in biological systems.
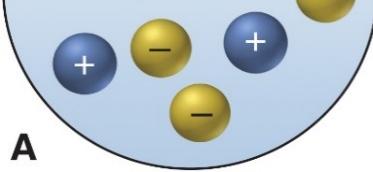
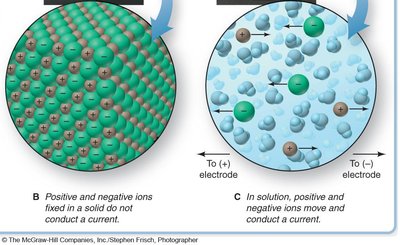
Solutions, Electrolytes, and Dissociation
Electrolytes and Dissociation
Electrolytes are substances that dissociate into ions when dissolved in water, allowing the solution to conduct electricity. The process of dissociation separates the ions, which can move freely in solution.
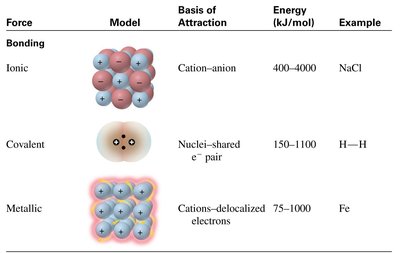
Summary Table: Types of Chemical Bonds
Force | Model | Basis of Attraction | Energy (kJ/mol) | Example |
|---|---|---|---|---|
Ionic | Cation-anion | Cation-anion | 400–4000 | NaCl |
Covalent | Nuclei-shared e- pair | Nuclei-shared e- pair | 150–1100 | H–H |
Metallic | Cations-delocalized electrons | Cations-delocalized electrons | 75–1000 | Fe |
Additional info: Academic context was added to clarify definitions, equations, and relationships between concepts, and to ensure completeness for exam preparation.
