 Back
BackEntropy Change During Vaporization of Methanol
Study Guide - Smart Notes

Q4. What is the change in entropy that occurs in the system when 1.00 mol of methanol (CH3OH) vaporizes from a liquid to a gas at its boiling point (64.6°C)? For methanol, ΔHvap = 35.2 kJ/mol.
Background
Topic: Thermodynamics – Entropy Change During Phase Transitions
This question tests your understanding of how to calculate the entropy change () for a substance undergoing a phase change (liquid to gas) at its boiling point. The process is isothermal, and the heat absorbed is equal to the enthalpy of vaporization.
Key Terms and Formulas
Entropy (): A measure of disorder or randomness in a system.
Enthalpy of vaporization (): The energy required to convert 1 mole of a liquid to a gas at constant temperature and pressure.
Boiling point: The temperature at which the liquid turns into gas (here, 64.6°C).
Formula for entropy change during a phase transition:
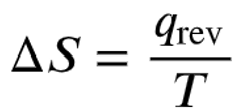
Where is the heat absorbed in a reversible process (here, ), and is the absolute temperature in Kelvin.
Step-by-Step Guidance
Convert the boiling point from Celsius to Kelvin:
Express the enthalpy of vaporization in joules (since entropy is typically in J/K):
Plug the values into the entropy change formula:
Set up the calculation with your converted values, but do not solve yet:
Try solving on your own before revealing the answer!
Final Answer: 98.2 J/mol·K
The entropy change is positive, reflecting increased disorder as methanol vaporizes.
