 Back
BackEntropy, Spontaneity, and Gibbs Free Energy: A Study Guide
Study Guide - Smart Notes

The Concept of Entropy
Boltzmann’s View of Entropy
Entropy (S) is a thermodynamic property that quantifies the degree of disorder or randomness in a system. Boltzmann’s view connects entropy to the number of microscopic configurations (microstates, W) available to a system:
Microstates: Specific arrangements of particles among energy levels.
Boltzmann Equation:
Boltzmann constant (kB): Relates entropy to the number of microstates; .
As the number of accessible microstates increases (by increasing energy or volume), entropy increases.
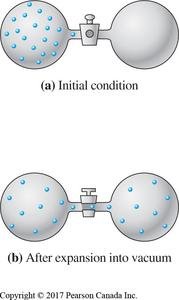
Example: Expansion of a gas into a vacuum increases the number of accessible microstates, thus increasing entropy.
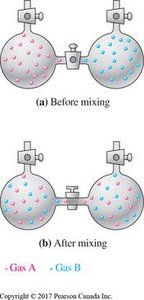
Example: Mixing two different gases increases the number of microstates and thus the entropy of the system.
Clausius’ View of Entropy
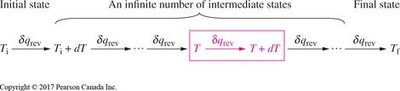
Clausius defined entropy change in terms of heat transfer in a reversible process:
Mathematical Definition:
For a finite process:
For a reversible isothermal process:
Evaluating Entropy and Entropy Changes
Entropy Changes in Physical Processes
Entropy increases in processes where disorder increases. Four common situations that increase entropy:
Formation of liquids from solids
Formation of gases from solids or liquids
Increase in the number of gas molecules in a reaction
Increase in temperature
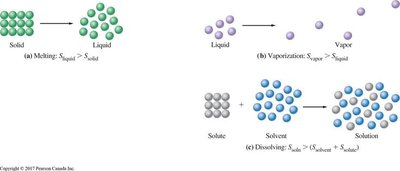
Example: Melting, vaporization, and dissolving all increase entropy.
Qualitative Predictions of Entropy Change
Predicting whether a process increases or decreases entropy involves considering the number of particles, phase changes, and temperature changes.

Entropy Change for Phase Transitions
During a phase transition at constant temperature, the entropy change is given by:

Entropy Change for Heating or Cooling
When a substance is heated or cooled at constant pressure:

Entropy Change for Ideal Gases
For an ideal gas undergoing isothermal expansion or compression:
Summary Table: Enthalpy and Entropy Changes
Process | Enthalpy Change | Entropy Change |
|---|---|---|
Phase transition at transition temperature | ||
Heating/cooling at constant pressure | ||
Change in state for ideal gas |

Calculating Standard Entropy of Reaction
The standard entropy change for a reaction is calculated using standard molar entropies:

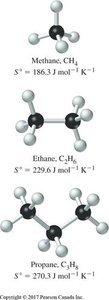
Entropy and Molecular Complexity
Standard molar entropy increases with molecular complexity (more atoms per molecule):
Criteria for Spontaneous Change: The Second Law of Thermodynamics
The Second Law of Thermodynamics
The second law states that the entropy of the universe increases for all spontaneous processes:
Spontaneity criteria:
: spontaneous
: nonspontaneous
: reversible
Standard Gibbs Energy Change, ∆G
Gibbs Free Energy and Spontaneity
Gibbs free energy (G) is a thermodynamic potential that predicts the spontaneity of a process at constant temperature and pressure:
If , the process is spontaneous.
If , the process is nonspontaneous.
If , the process is at equilibrium.
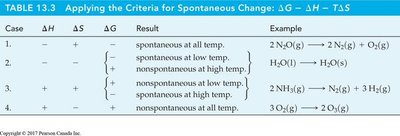
Applying the Criteria for Spontaneous Change
Case | ΔH | ΔS | ΔG | Result | Example |
|---|---|---|---|---|---|
1 | - | + | - | Spontaneous at all temp. | |
2 | - | - | +/− | Spontaneous at low temp. | |
3 | + | + | +/− | Spontaneous at high temp. | |
4 | + | - | + | Nonspontaneous at all temp. |

Calculating ΔG for a Reaction
ΔG can be calculated from enthalpy and entropy changes:

Gibbs Energy Change and Equilibrium
Relationship Between ΔG and the Equilibrium Constant (K)
At equilibrium, the standard Gibbs energy change is related to the equilibrium constant:
If , the reaction proceeds forward (spontaneous).
If , the reaction proceeds in reverse (nonspontaneous).
If , the system is at equilibrium.

Predicting the Direction of Spontaneous Chemical Change
ΔrG | Spontaneous Reaction |
|---|---|
< 0 | Left to right (→) |
> 0 | Right to left (←) |
= 0 | Equilibrium (⇌) |
ΔG° and K as Functions of Temperature
Temperature Dependence of Equilibrium Constant
The van’t Hoff equation relates the equilibrium constant to temperature:
For two temperatures:

Worked Example: Relating Equilibrium Constants and Temperature

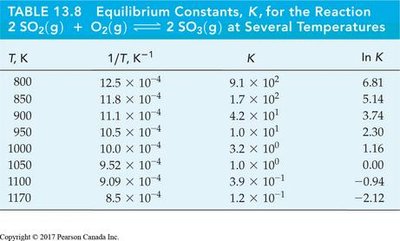
Summary Table: Equilibrium Constants at Different Temperatures
T, K | 1/T, K-1 | K | ln K |
|---|---|---|---|
800 | 12.5 × 10-4 | 9.1 × 102 | 6.81 |
850 | 11.8 × 10-4 | 1.7 × 102 | 5.14 |
900 | 11.1 × 10-4 | 4.2 × 101 | 3.74 |
950 | 10.5 × 10-4 | 1.0 × 101 | 2.30 |
1000 | 10.0 × 10-4 | 2.3 × 100 | 0.83 |
1050 | 9.52 × 10-4 | 5.0 × 10-1 | -0.69 |
1100 | 9.09 × 10-4 | 1.1 × 10-1 | -2.20 |
1170 | 8.5 × 10-4 | 1.2 × 10-1 | -2.12 |
Additional info: This guide covers the core concepts of entropy, spontaneity, and Gibbs free energy, including their calculation, interpretation, and application to chemical and physical processes. Worked examples and tables are included to reinforce understanding and provide practical calculation strategies.
