 Back
BackEquilibrium Constants and Heterogeneous Equilibria: General Chemistry Study Guide
Study Guide - Smart Notes

Equilibrium Constants and Heterogeneous Equilibria
Introduction to Equilibrium Constants
Chemical equilibrium occurs when the rates of the forward and reverse reactions are equal, resulting in constant concentrations of reactants and products. The equilibrium constant quantifies the ratio of product and reactant concentrations at equilibrium.
Equilibrium Constant (K): A dimensionless value that expresses the ratio of concentrations (or partial pressures) of products to reactants at equilibrium.
Kc: Uses concentrations in molarity (M).
Kp: Uses partial pressures in atmospheres (atm).
Unitless Nature: Equilibrium constants are unitless because concentrations and pressures are expressed as activities, which are dimensionless quantities.
Activities and Approximations
In equilibrium calculations, activities are used to represent the effective concentration of a species. For ideal gases and solutions, activities can be approximated by partial pressures (in atm) and concentrations (in M), respectively.
Ideal Gas: Activity =
Solute in Ideal Solution: Activity =
Pure Solid or Liquid: Activity = 1
Substance | Activity | Simplified Form |
|---|---|---|
Ideal gas | P (atm) | |
Solute in ideal solution | [C] (M) | |
Pure solid or liquid | 1 | 1 |
Heterogeneous Equilibria
Heterogeneous equilibria involve reactants and products in different phases (solid, liquid, gas). The activities of pure solids and liquids are always 1, so they are omitted from equilibrium constant expressions.
Equilibrium Expression: Only includes species whose concentrations or pressures can change during the reaction.
Solvents: If a reactant or product is also the solvent, it is omitted from the equilibrium expression.
Example: For the reaction , the equilibrium constant is .
Equilibrium Constant Expressions
The equilibrium constant expression is derived from the balanced chemical equation. For a general reaction:
The equilibrium constant is:
Only include species whose concentrations/pressures change.
Solids and pure liquids are omitted.
Using RICE (ICE) Tables for Equilibrium Calculations
RICE (Reaction, Initial, Change, Equilibrium) tables are powerful tools for tracking concentration changes and solving equilibrium problems.
Step 1: List initial concentrations or partial pressures.
Step 2: Use stoichiometry to determine changes (expressed as x).
Step 3: Write equilibrium concentrations in terms of x.
Step 4: Substitute into the equilibrium constant expression and solve for x.
Step 5: Calculate final equilibrium concentrations.
Example: NO and H2 Equilibrium
For the reaction , given initial concentrations and one equilibrium concentration, use the RICE table to solve for the equilibrium constant.
Reagent | Initial (M) | Change | Equilibrium (M) |
|---|---|---|---|
NO | 0.100 | -2x | 0.062 |
H2 | 0.050 | -2x | 0.012 |
N2 | 0 | +x | 0.019 |
H2O | 0.100 | +2x | 0.138 |
Equilibrium constant expression:
Plug in values to solve for .
Quadratic Formula in Equilibrium Calculations
When the equilibrium constant expression leads to a quadratic equation, use the quadratic formula:
Neglecting x: If x is much smaller than the initial concentration or pressure (typically less than 5%), it can be neglected for simplification.
Example: Methane and Iodine Equilibrium
For the reaction , use the ICE table and quadratic formula to solve for equilibrium partial pressures.
Reactant | Initial (atm) | Change | Equilibrium (atm) |
|---|---|---|---|
CH4 | 0.138 | -x | 0.136 |
I2 | 0.105 | -x | 0.103 |
CH3I | 0 | +x | 0.00178 |
HI | 0 | +x | 0.00178 |
Equilibrium constant expression:
Plug in values and solve for x using the quadratic formula.
Summary of Key Concepts
Equilibrium constants are unitless because they are based on activities.
Solids and pure liquids are omitted from equilibrium expressions.
RICE tables are essential for solving equilibrium problems.
Quadratic equations may be required for more complex equilibrium calculations.
Practice Problem Example: Bromine and Chlorine Equilibrium
For the reaction , use the ICE table and equilibrium constant to solve for equilibrium partial pressures.
Reactant | Initial (atm) | Change | Equilibrium (atm) |
|---|---|---|---|
Br2 | 0 | +x | x |
Cl2 | 0 | +x | x |
BrCl | 1.00 | -2x | 1.00-2x |
Equilibrium constant expression:
Solve for x and determine equilibrium partial pressures.
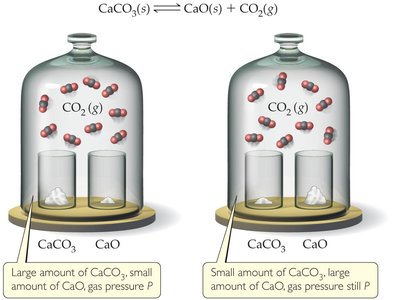
Visual Representation of Solid-Gas Equilibrium
Solid-gas equilibria are not affected by the amount of solid present, as long as some is present. The gas pressure at equilibrium remains constant regardless of the amount of solid.

Example: The equilibrium pressure of CO2 is the same whether there is a large or small amount of CaCO3 present, as long as equilibrium is established.
Conclusion
Understanding equilibrium constants, heterogeneous equilibria, and the use of RICE tables is fundamental for solving equilibrium problems in general chemistry. Mastery of these concepts enables students to analyze and predict the behavior of chemical systems at equilibrium.
