 Back
BackEssential General Chemistry Exam 1 Study Guide: Atoms, Quantum Mechanics, and Measurement
Study Guide - Smart Notes

Conversions and Calculations in Chemistry
Grams, Moles, and Number of Atoms
Understanding the relationships between mass, moles, and the number of atoms is fundamental in chemistry. These conversions allow chemists to relate measurable quantities to the atomic scale.
Grams to Moles: Use the molar mass (g/mol) of a substance to convert grams to moles.
Moles to Number of Atoms: Multiply moles by Avogadro's number ( atoms/mol).
Grams to Moles to Number of Atoms: Combine the above steps for multi-step conversions.
Example: If you have 10 g of carbon, divide by the molar mass (12.01 g/mol) to get moles, then multiply by Avogadro's number for atoms.
Fundamental Laws of Chemistry
Law of Conservation of Mass, Definite Proportions, and Multiple Proportions
These laws describe how matter behaves in chemical reactions and how elements combine to form compounds.
Law of Conservation of Mass: Mass is neither created nor destroyed in a chemical reaction.
Law of Definite Proportions: A compound always contains the same proportion of elements by mass.
Law of Multiple Proportions: When two elements form more than one compound, the ratios of the masses of the second element that combine with a fixed mass of the first element are simple whole numbers.
Example: Calculating mass ratios in compounds using sample data.
Atomic Structure and Periodic Table
Atomic Number, Atomic Mass, and Molar Mass
The periodic table provides essential information about each element, including its atomic number, atomic mass, and molar mass.
Atomic Number (Z): Number of protons in the nucleus.
Atomic Mass: Weighted average mass of an element's isotopes.
Molar Mass: Mass of one mole of an element (g/mol).
Example: Identifying these values for sodium (Na): Z = 11, atomic mass ≈ 22.99, molar mass = 22.99 g/mol.
Quantum Mechanics and Atomic Models
De Broglie’s Equation
De Broglie proposed that particles such as electrons have wave-like properties. The wavelength of a particle is given by:
Equation:
Where: = wavelength, = Planck's constant, = mass, = velocity.
Example: Calculating the wavelength of an electron moving at a certain speed.
Weighted Average of Atomic Masses (Isotopes)
The atomic mass of an element is a weighted average based on the abundance and mass of its isotopes.
Equation:
Example: If an element has two isotopes, calculate the average using their masses and abundances.
Bohr’s Model for the Hydrogen Atom
Bohr's model explains the quantized energy levels of electrons in hydrogen. Energy changes occur when electrons move between levels.
Energy Change Equation:
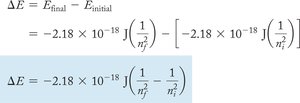
Where: = final energy level, = initial energy level.
Application: Calculate the energy required to excite or relax an electron and the wavelength of light involved.
Wave Properties and Criteria
Definitions and Units
Waves are characterized by several properties:
Wavelength (): Distance between two consecutive peaks (meters).
Frequency (): Number of cycles per second (Hz).
Amplitude: Height of the wave (meters).
Example: Light waves have wavelengths in nanometers (nm).
Conversion Factors and Significant Figures
Using Conversion Factors
Conversion factors are used to change units and quantities in chemistry, often in multi-step calculations.
Example: Converting cm to m, then m to km using appropriate factors.
Significant Figures
Significant figures reflect the precision of measurements and must be correctly reported in calculations.
Rules: Nonzero digits are always significant; zeros are significant depending on their position.
Example: Reporting the answer to the correct number of significant figures after multiplication or division.
Quantum Numbers and Their Significance
Determining Quantum Numbers
Quantum numbers describe the properties of electrons in atoms:
Principal Quantum Number (): Energy level (positive integer).
Angular Momentum Quantum Number (): Shape of orbital ().
Magnetic Quantum Number (): Orientation ().
Example: For , can be 0, 1, or 2; for , can be -1, 0, or 1.
Information from Quantum Numbers
Each quantum number provides specific information about the electron:
: Energy and size of orbital.
: Shape of orbital.
: Orientation in space.
Example: , describes a 2s orbital.
Photon Energy and Photoelectric Effect
Energy of a Photon
The energy of a photon is related to its frequency and wavelength:
Equation:
Where: = energy, = Planck's constant, = frequency, = speed of light, = wavelength.
Application: Calculate energy for one photon, multiple photons, or a mole of photons.
Photoelectric Effect
Einstein's model describes the ejection of electrons from a metal when exposed to light:
Equation:
Application: Calculate the kinetic energy of electrons using photon energy and binding energy.
Heisenberg Uncertainty Principle
Significance and Equation
The Heisenberg uncertainty principle states that it is impossible to simultaneously know the exact position and momentum of a particle.
Equation:
Significance: Sets a fundamental limit on measurement precision in quantum systems.
Additional info: Academic context and examples were added to ensure completeness and clarity for exam preparation.
