Back
BackEssential Polyatomic Ions for General Chemistry
Study Guide - Smart Notes

Polyatomic Ions
Introduction to Polyatomic Ions
Polyatomic ions are charged species composed of two or more atoms covalently bonded, acting as a single unit in chemical reactions. Understanding their names, formulas, and charges is fundamental for predicting compound formation, writing chemical equations, and performing stoichiometric calculations in general chemistry.
Definition: A polyatomic ion is a group of atoms with an overall charge, held together by covalent bonds.
Importance: Polyatomic ions frequently appear in ionic compounds, acid-base reactions, and solution chemistry.
Notation: The charge is shown as a superscript (e.g., SO42−).
Common Polyatomic Ions
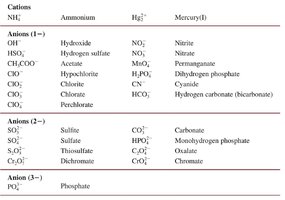
The following table summarizes the most important polyatomic ions, grouped by charge and type. Memorizing these ions is essential for success in general chemistry.
Cations | Anions (1−) | Anions (2−) | Anion (3−) |
|---|---|---|---|
NH4+ (Ammonium) Hg22+ (Mercury(I)) | OH− (Hydroxide) HSO4− (Hydrogen sulfate) CH3COO− (Acetate) ClO− (Hypochlorite) ClO2− (Chlorite) ClO3− (Chlorate) ClO4− (Perchlorate) NO2− (Nitrite) NO3− (Nitrate) MnO4− (Permanganate) H2PO4− (Dihydrogen phosphate) CN− (Cyanide) HCO3− (Hydrogen carbonate/bicarbonate) | SO32− (Sulfite) SO42− (Sulfate) S2O32− (Thiosulfate) CrO42− (Chromate) CO32− (Carbonate) HPO42− (Monohydrogen phosphate) C2O42− (Oxalate) Cr2O72− (Dichromate) | PO43− (Phosphate) |
Key Properties and Examples
Charge: Polyatomic ions can be positive (cations) or negative (anions), with charges ranging from 1− to 3−.
Compound Formation: Polyatomic ions combine with other ions to form ionic compounds, e.g., sodium sulfate (Na2SO4).
Acid-Base Chemistry: Many polyatomic ions are conjugate bases of acids, e.g., carbonate (CO32−) from carbonic acid.
Example: Naming Ionic Compounds with Polyatomic Ions
Potassium nitrate: KNO3 (contains K+ and NO3−)
Calcium phosphate: Ca3(PO4)2 (contains Ca2+ and PO43−)
Formulas and Charges
To write formulas, balance the total positive and negative charges.
Example equation for charge balance: