 Back
BackEssentials: Units, Measurement, and Problem Solving – Mini Study Guide
Study Guide - Smart Notes

Essentials: Units, Measurement, and Problem Solving
Introduction to Measurement and Units
Measurement is fundamental to chemistry, providing quantitative data for scientific analysis. Every measurement consists of a numerical value and a unit, which together convey both the magnitude and the dimension of the quantity measured.
Numerical Value: Indicates the precision of the measurement instrument.
Unit: Specifies the dimension (e.g., meters for length, kilograms for mass).
Systems of Units: The metric system is used globally, while the English system is primarily used in the U.S. Scientists use the International System of Units (SI), which is based on the metric system.
Example: 5.9 m (meters), 3.7 kg (kilograms), 25.0 cm (centimeters)
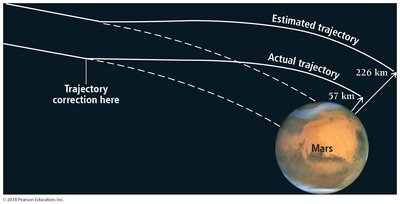
Example: The Mars Climate Orbiter failed due to a unit conversion error between metric and English units, highlighting the importance of correct unit usage.
Metric System: Prefix Multipliers
The metric system uses prefix multipliers to represent quantities larger or smaller than the base unit. Understanding these prefixes is essential for converting between units.
Common Prefixes: kilo-, centi-, milli-, micro-, nano-, pico-
Conversion Factors: 1 km = 1000 m, 100 cm = 1 m, 1000 mm = 1 m, 10 mm = 1 cm
Application: Prefixes apply to meters (m), grams (g), and liters (l).
Measurement of Length, Volume, and Mass
Length, volume, and mass are fundamental physical quantities in chemistry. The SI unit for length is the meter, for volume is the liter or cubic centimeter, and for mass is the kilogram.
Length: Meter (m) is slightly longer than a yard.
Volume: 1 mL = 1 cm3. Common units: liters (L), milliliters (mL), cubic centimeters (cm3).
Mass: Measured in grams (g) or kilograms (kg).


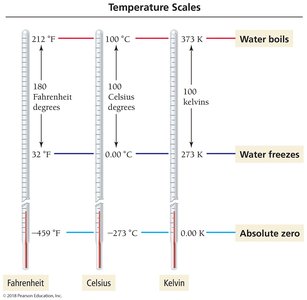
Temperature Scales and Conversions
Temperature is measured using three main scales: Celsius, Fahrenheit, and Kelvin. Converting between these scales is a common task in chemistry.
Celsius (°C): Water freezes at 0°C, boils at 100°C.
Fahrenheit (°F): Water freezes at 32°F, boils at 212°F.
Kelvin (K): Absolute zero is 0 K; water freezes at 273 K, boils at 373 K.
Conversion Equations:
Example: 37.00°C + 273.15 = 310.15 K; 77 K – 273.15 = –196°C
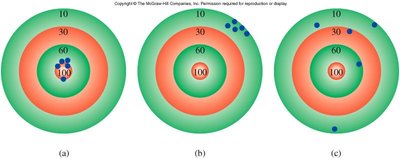
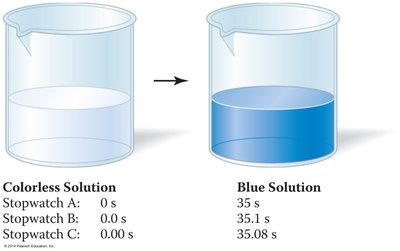
Accuracy, Precision, and Reliability of Measurements
Understanding the reliability of measurements is crucial. Accuracy refers to how close a measurement is to the true value, while precision refers to how close repeated measurements are to each other.
Accurate and Precise: Measurements are both close to the true value and to each other.
Precise but Not Accurate: Measurements are close to each other but not to the true value.
Neither Accurate nor Precise: Measurements are scattered and not close to the true value.
Significant Figures
Significant figures (sig figs) express the uncertainty in a measurement. The rules for counting significant figures are essential for proper reporting and calculation.
Rule 1: All non-zero digits are significant.
Rule 2: Zeros between non-zero digits are significant.
Rule 3: Leading zeros are not significant.
Rule 4: Trailing zeros are significant if there is a decimal point.
Scientific Notation: Only the coefficient is counted for significant figures.
Exact Numbers: Numbers from definitions or counting objects have infinite significant figures.
Significant Figures in Calculations
When performing calculations, the number of significant figures in the result depends on the operation:
Addition/Subtraction: The result has the same number of decimal places as the measurement with the fewest decimal places.
Multiplication/Division: The result has the same number of significant figures as the factor with the fewest significant figures.
Scientific Notation
Scientific notation is used to express very large or very small numbers conveniently. It is written as , where N is a number between 1 and 10, and n is an integer.
Example: (number of atoms in 12 g of carbon)
Example: (mass of a single carbon atom in grams)
Density: An Intensive Property
Density is defined as mass per unit volume and is an intensive property, meaning it does not depend on the amount of substance.
Formula:
Units: g/cm3, kg/m3
Example: Mercury has a density of 13.6 g/cm3. The mass of 95 mL of mercury is g.
Energy and Its Units
Energy is the capacity to do work. It is classified as kinetic (movement) or potential (position/composition). The First Law of Thermodynamics states that energy is conserved.
Kinetic Energy:
Potential Energy: Associated with position or composition.
Units: Joule (J), calorie (cal), kilocalorie (kcal), kilojoule (kJ), kilowatt-hour (kWh)
Conversions: 1 cal = 4.184 J, 1 kcal = 1000 cal = 4184 J, 1 kWh = J

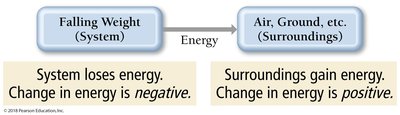
Energy Terminology: System and Surroundings
In thermodynamics, the system is the part under study, and the surroundings are everything else. The universe is the sum of system and surroundings.
Energy Transfer: Energy can flow between system and surroundings.
Exothermic: System loses energy; surroundings gain energy.
Endothermic: System gains energy; surroundings lose energy.
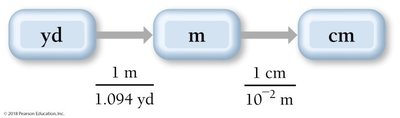
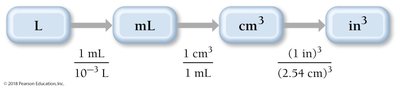
Dimensional Analysis: Unit Conversion
Dimensional analysis is a systematic method for converting between units using conversion factors. It is essential for solving chemistry problems involving measurements.
Conversion Factor: A ratio expressing the relationship between two units (e.g., 1 inch = 2.54 cm).
Strategy: Sort information, devise a plan, solve, and check the answer.
Example: 2.00 ft × (12 in/1 ft) = 24.0 in
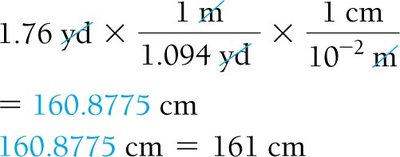
Compound Unit Conversions
Compound units involve converting units raised to a power or combining multiple unit conversions, such as speed (m/s) or volume (cm3).
Example: 55 mi/hr to m/s: m/s
Example: 105 km/hr to m/s: m/s
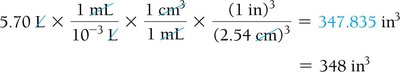
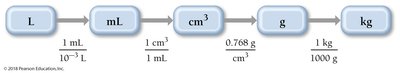
Density as a Conversion Factor
Density can be used to convert between mass and volume. This is especially useful in practical chemistry problems.
Formula:
Example: To find the mass of jet fuel:
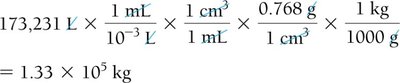
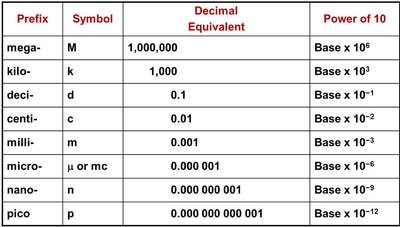
Summary Table: Metric Prefixes
The following table summarizes common metric prefixes used in chemistry:
Prefix | Symbol | Decimal Equivalent | Power of 10 |
|---|---|---|---|
mega- | M | 1,000,000 | Base x 106 |
kilo- | k | 1,000 | Base x 103 |
deci- | d | 0.1 | Base x 10-1 |
centi- | c | 0.01 | Base x 10-2 |
milli- | m | 0.001 | Base x 10-3 |
micro- | μ or mc | 0.000 001 | Base x 10-6 |
nano- | n | 0.000 000 001 | Base x 10-9 |
pico- | p | 0.000 000 000 001 | Base x 10-12 |
Additional info: This study guide covers all foundational aspects of units, measurement, and problem solving as outlined in Chapter E of a general chemistry course. It includes definitions, examples, formulas, and step-by-step strategies for unit conversions and calculations, making it suitable for exam preparation and practical laboratory work.
