 Back
BackExam 3 Review: Thermochemistry, Electrochemistry, and Titrations
Study Guide - Smart Notes

Thermochemistry
Key Concepts in Thermochemistry
Thermochemistry studies the energy and heat changes involved in chemical reactions. Understanding the relationships between enthalpy, entropy, and free energy is essential for predicting reaction spontaneity and equilibrium.
Enthalpy (ΔH): The heat content of a system at constant pressure. Exothermic reactions have negative ΔH; endothermic reactions have positive ΔH.
Entropy (ΔS): A measure of disorder or randomness in a system. Higher entropy means greater disorder. Entropy increases with temperature, volume, and the number of particles or microstates.
Gibbs Free Energy (ΔG): Determines the spontaneity of a process. A negative ΔG indicates a spontaneous process.
Spontaneous Process: Occurs without external intervention. Spontaneity depends on ΔH, ΔS, and temperature.
Reversible vs. Irreversible Processes: Reversible processes can return to their original state without net energy change; irreversible processes cannot.
Boltzmann Equation: Relates entropy to the number of microstates (W):
Physical State and Entropy: Gases have higher entropy than liquids, which have higher entropy than solids.
Standard Molar Entropy: Tabulated values for substances at standard conditions (1 bar, 298 K).
Calculating Entropy Changes:
Gibbs Free Energy Equation:
Equilibrium and Free Energy:
Nonstandard Free Energy:
Example: Calculating the temperature at which a reaction becomes spontaneous: Set and solve for using and .
Example: For the reaction , J/mol·K (using standard entropy values).
Example: For the reaction , kJ/mol.
Example: Calculating for a phase change:

Example: Calculating :

Example: Calculating the equilibrium constant from :
Given kJ/mol at 298 K,
Example: Calculating under nonstandard conditions:
Titrations
Key Concepts in Titrations
Titration is a technique used to determine the concentration of an unknown solution by reacting it with a solution of known concentration. Titration curves plot pH versus volume of titrant added and reveal important points such as the equivalence point and buffer region.
Titration Curve: Graph of pH versus volume of titrant added. Key regions include the initial region, buffer region, equivalence point, and over-titration region.
Strong/Strong Titration: Both acid and base are strong; pH changes rapidly near equivalence.
Strong/Weak Titration: One reactant is weak; buffer region is present, and equivalence point pH is not neutral.
Calculating pH at Various Points:
Before titrant is added: Use initial concentration and dissociation constant (Ka or Kb).
Buffer region: Use Henderson-Hasselbalch equation.
At equivalence: Only the conjugate base or acid is present; calculate pH from hydrolysis.
After equivalence: Excess titrant determines pH.
Example: Titration of 0.150 M formic acid (Ka = ) with 0.150 M NaOH (50.00 mL sample):
Stage | pH |
|---|---|
Before NaOH added | 2.28 |
After 10.00 mL NaOH | 3.14 |
At half-equivalence | 3.74 |
At equivalence | 8.81 |
After 60.00 mL NaOH | 12.13 |
Example: Calculating the initial concentration of a weak acid (HA) titrated with 0.150 M NaOH, equivalence at 42.60 mL for a 35.00 mL sample: M.
Example: If pH at equivalence is 8.72, .
Example: Titration of 0.200 M methylamine (Kb = ) with 0.200 M HCl (40.00 mL sample):
Stage | pH |
|---|---|
Initially | 11.97 |
After 20.00 mL HCl | 10.64 |
At equivalence | 5.76 |
After 50.00 mL HCl | 1.65 |
Electrochemistry
Key Concepts in Electrochemistry
Electrochemistry deals with the interconversion of chemical and electrical energy. It involves redox reactions, cell potentials, and the operation of batteries and electrolysis cells.
Oxidation Numbers: Assigned to atoms to track electron transfer. Common values: O is usually –2, H is +1, etc.
Oxidation and Reduction: Oxidation is loss of electrons (OIL), reduction is gain of electrons (RIG). "LEO the lion goes GER" is a mnemonic.
Oxidizing and Reducing Agents: The oxidizing agent is reduced; the reducing agent is oxidized.
Balancing Redox Reactions: Split into half-reactions, balance atoms and charges, combine.
Voltaic (Galvanic) Cell: Converts chemical energy to electrical energy. Key parts: anode (oxidation), cathode (reduction), salt bridge, external circuit.
Cell Potential (E0): Calculated from standard reduction potentials.
Standard Reduction Potentials: Tabulated values used to determine cell potential and reaction direction.
Strength of Agents: Higher (more positive) reduction potential = stronger oxidizing agent; lower (more negative) = stronger reducing agent.
Relationship to Free Energy and Equilibrium: and
Nonstandard Cell Potentials: at 25°C
Batteries and Fuel Cells: Practical applications of voltaic cells.
Electrolysis: Uses electrical energy to drive nonspontaneous reactions. Amount of substance produced/consumed is related to current and time.
Example: Assigning oxidation numbers: Xe in XeF4 is +4; I in Mg(IO3)2 is +5.
Example: Balancing redox reactions:
Example: Calculating cell potential for Co2+/Co and Au3+/Au:
V
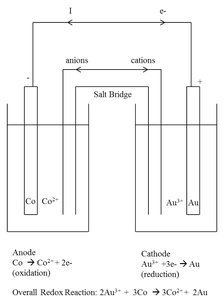
Example: Calculating standard free energy for the cell:
For , C/mol, V: kJ/mol
Example: Calculating equilibrium constant:
For kJ/mol, (very large)
Example: Calculating emf under nonstandard conditions:
For [Co2+] = 0.30 M, [Au3+] = 0.0010 M, V
Example: Calculating mass of Co consumed by a current:
Current (I) × time (t) = charge (Q)
Q / F = moles of e–
Stoichiometry: moles of e– to moles of Co
Moles of Co × molar mass = grams Co
For 1 mA over 3,000 hours: 3.30 g Co consumed
