 Back
BackExpressing Equilibrium Constants for Gas-Phase Reactions
Study Guide - Smart Notes

Q7. Express the equilibrium constant for the following reaction:
2 CH3Cl(g) + Cl2(g) ⇔ 2 CH2Cl2(g) + H2(g)
Background
Topic: Chemical Equilibrium – Equilibrium Constant Expressions
This question tests your ability to write the equilibrium constant expression (KP) for a gas-phase reaction, using partial pressures of the reactants and products.
Key Terms and Formulas
Equilibrium constant (KP): A ratio of the partial pressures of products to reactants, each raised to the power of their stoichiometric coefficients.
Partial pressure (P): The pressure exerted by a single gas in a mixture.
General formula:
Step-by-Step Guidance
Write the balanced chemical equation:
Identify the products and reactants, and their stoichiometric coefficients:
Products: CH2Cl2 (2), H2 (1)
Reactants: CH3Cl (2), Cl2 (1)
Set up the equilibrium constant expression using partial pressures:
Check the image options for the correct mathematical form. Look for the expression that matches the formula above.
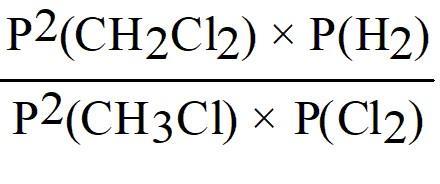
Try solving on your own before revealing the answer!
Final Answer:
This matches the mathematical form shown in image_2. The equilibrium constant expression is constructed by raising each partial pressure to the power of its coefficient in the balanced equation.
