 Back
BackFree Energy and Thermodynamics: Entropy, Spontaneity, and Gibbs Free Energy
Study Guide - Smart Notes

Free Energy and Thermodynamics
Introduction to Thermodynamics and Spontaneity
Thermodynamics is the study of energy changes in chemical processes. It predicts whether a process will occur under given conditions, focusing on the concepts of energy, entropy, and spontaneity. Spontaneous processes occur without ongoing outside intervention, while nonspontaneous processes require energy input.
First Law of Thermodynamics: Energy cannot be created or destroyed; it is conserved in all processes.
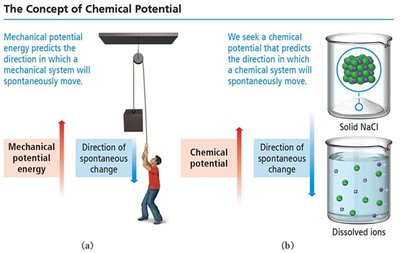
Spontaneity: Determined by comparing the chemical potential energy before and after a reaction. If the system's potential energy decreases, the process is thermodynamically favorable.
Spontaneity vs. Rate: A spontaneous process may be slow or fast; spontaneity does not imply speed.
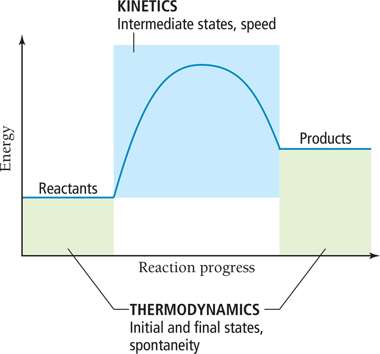
Kinetics Versus Thermodynamics
Thermodynamics determines if a reaction can occur, while kinetics determines how fast it occurs. The energy profile of a reaction shows the difference between the initial and final states (thermodynamics) and the pathway (kinetics).
Spontaneous and Non spontaneous Processes
Potential Energy and Spontaneity
Spontaneous processes often move from higher to lower potential energy (exothermic), but some endothermic processes are also spontaneous due to entropy changes.
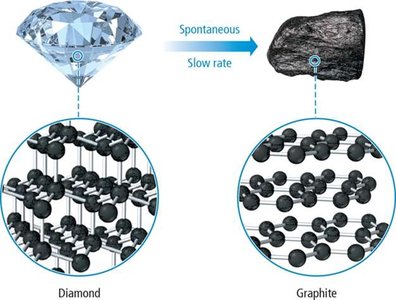
Example: The conversion of diamond to graphite is spontaneous but extremely slow.
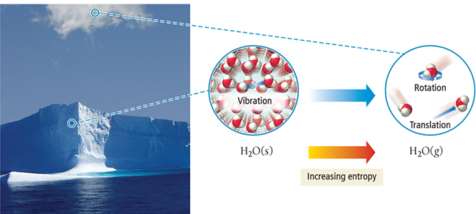
Entropy and State Changes
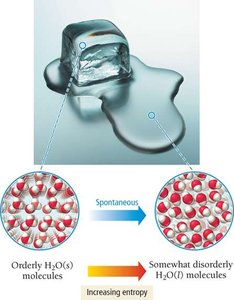
Entropy (S) is a measure of the disorder or randomness of a system. Processes that increase entropy are often spontaneous, even if they absorb energy (endothermic).
Melting Ice: Melting is endothermic but spontaneous above 0°C due to increased entropy.
Evaporation and Dissolution: Both processes increase entropy as particles become more disordered.
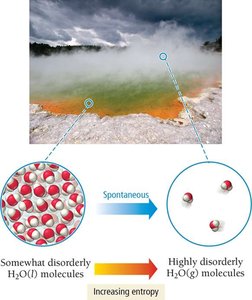
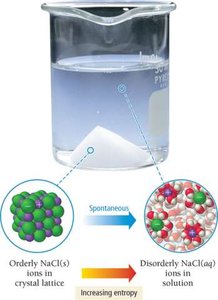
Entropy: The Second Law of Thermodynamics
Definition and Statistical Interpretation
The second law of thermodynamics states that for any spontaneous process, the entropy of the universe increases (ΔSuniv > 0). Entropy is a state function and can be described statistically:
Boltzmann Equation: where k is the Boltzmann constant ( J/K) and W is the number of microstates.
Microstate: A specific arrangement of particles and energy.
Macrostate: The overall state defined by measurable properties (P, V, T).
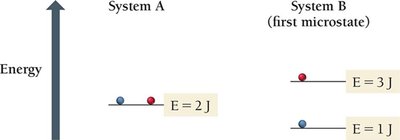
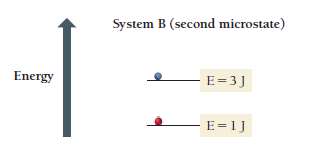
Macrostates, Microstates, and Probability
The macrostate with the highest entropy has the greatest number of microstates and is the most probable.
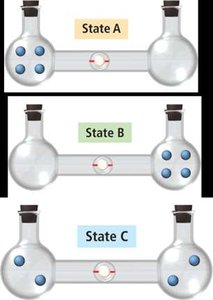
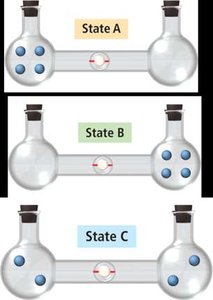
Entropy Change in State Change
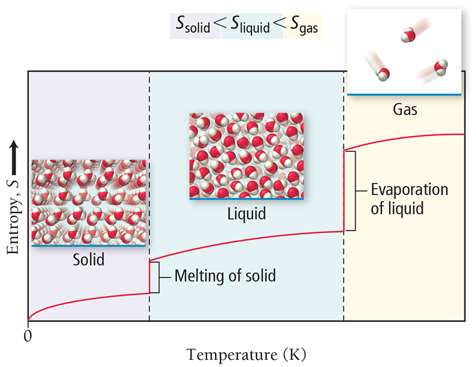
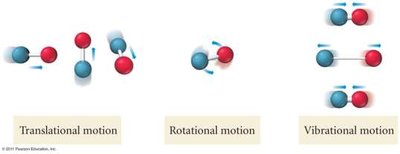
When a substance changes state, the number of possible microstates changes. Gases have the highest entropy, followed by liquids, then solids.
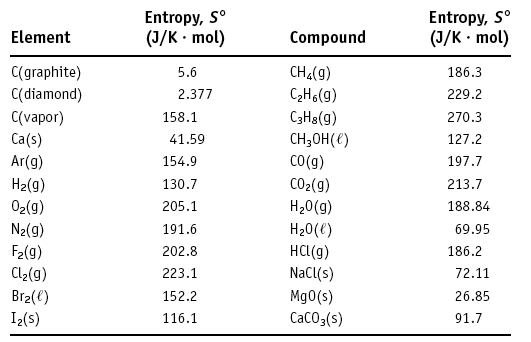
Standard Molar Entropy and Calculations
Standard State and Molar Entropy
The standard molar entropy (S°) is the entropy of one mole of a substance in its standard state (1 atm, 1 M, 25°C). Unlike enthalpy, the standard molar entropy of elements is not zero.
Units: J/(mol·K)
Third Law of Thermodynamics: The entropy of a perfect crystal at absolute zero is zero.
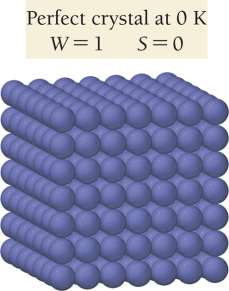
Factors Affecting Entropy
Molar Mass: Larger molar mass = higher entropy.
Allotropes: Less constrained structures (e.g., graphite vs. diamond) have higher entropy.
Physical State: Sgas > Sliquid > Ssolid
Molecular Complexity: More complex molecules have higher entropy.
Dissolution: Dissolved solids have higher entropy than undissolved solids.
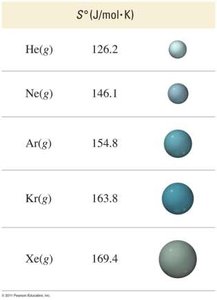



Calculating Entropy Change for a Reaction
The standard entropy change for a reaction is calculated as:
Entropy Change in the System and Surroundings
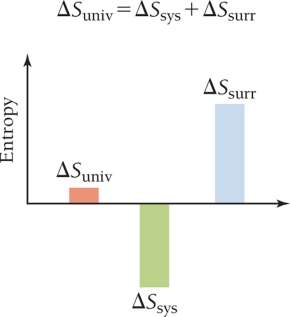
System, Surroundings, and Universe
For a process to be spontaneous, the total entropy change of the universe must be positive. If the system's entropy decreases, the surroundings must increase more to compensate.
Quantifying Entropy Changes in Surroundings
At constant pressure and temperature:
At constant temperature:
Gibbs Free Energy
Definition and Criteria for Spontaneity
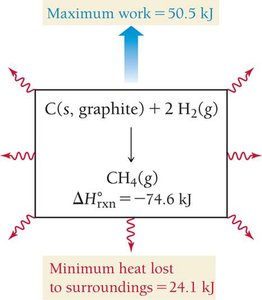
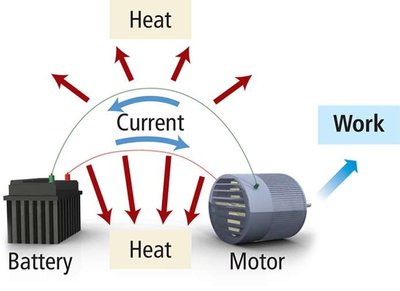
Gibbs free energy (G) is the maximum amount of work energy that can be released to the surroundings by a system at constant temperature and pressure. The change in Gibbs free energy determines spontaneity:
Spontaneous:
Nonspontaneous:
Equilibrium:
Calculating Gibbs Free Energy
For reactions at standard state:
Free Energy and Reversible Reactions
The change in free energy is the theoretical limit for the amount of work a reaction can do. Real reactions are usually irreversible due to energy lost as heat.

Gibbs Free Energy Under Nonstandard Conditions
When reactants and products are not in their standard states, the free energy change is:
Q: Reaction quotient
At equilibrium, and
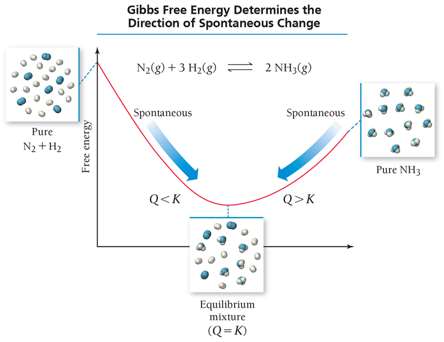
Relationship Between Free Energy and Equilibrium
The sign and magnitude of determine the direction of spontaneity and the value of the equilibrium constant (K):
If , (spontaneous forward)
If , (spontaneous reverse)
If , (equilibrium)
Temperature Dependence of K
The equilibrium constant depends on temperature, as shown by:
This equation predicts how K changes with temperature for a given reaction.
Summary Table: Standard Molar Entropies
Element | Entropy, S° (J/K·mol) | Compound | Entropy, S° (J/K·mol) |
|---|---|---|---|
C (graphite) | 5.6 | CH4(g) | 186.3 |
C (diamond) | 2.377 | C2H6(g) | 229.2 |
C (vapor) | 158.1 | C3H8(g) | 270.3 |
Ca (s) | 41.59 | CH3OH(l) | 127.2 |
Ar (g) | 154.9 | CO (g) | 197.7 |
H2(g) | 130.7 | CO2(g) | 213.7 |
O2(g) | 205.1 | H2O(g) | 188.84 |
N2(g) | 191.6 | H2O(l) | 69.95 |
F2(g) | 202.8 | HCl(g) | 186.2 |
Cl2(g) | 223.1 | NaCl(s) | 72.11 |
Br2(l) | 152.2 | MgO(s) | 26.85 |
I2(s) | 116.1 | CaCO3(s) | 91.7 |
