 Back
BackFree Energy and Thermodynamics: Study Notes for General Chemistry
Study Guide - Smart Notes

Free Energy and Thermodynamics
Introductory Concepts
Thermodynamics is the study of energy transformations, particularly the relationships between heat (q), work (w), and internal energy (ΔE). It provides the foundation for understanding spontaneity, equilibrium, and the direction of chemical processes.
Internal Energy (ΔE): The total energy contained within a system. It is a state function, meaning its value depends only on the state of the system, not the path taken to reach that state.
First Law of Thermodynamics: Energy cannot be created or destroyed, only transferred or converted. Expressed as:
Heat Capacity: or
Relationship between Enthalpy and Internal Energy:
Work at Constant Pressure:
Thermodynamics and Spontaneity
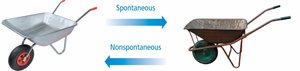
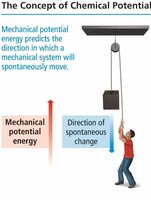
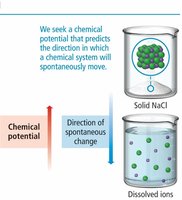
Spontaneity refers to the natural tendency of a process to occur without external intervention. Thermodynamics helps predict whether a reaction or process is spontaneous by comparing the potential energy of the system before and after the process.
Comparing Potential Energy
The direction of spontaneity can be determined by comparing the potential energy of the system at the start and the end. Systems tend to move toward lower potential energy, which is often associated with increased stability.
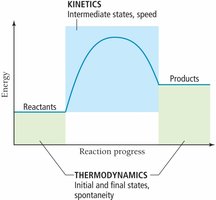
Thermodynamics vs. Kinetics
Thermodynamics determines whether a reaction is possible and the direction it will proceed, while kinetics describes the rate at which the reaction occurs. A process can be thermodynamically favorable but kinetically slow.
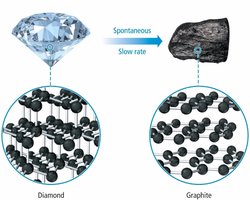
Thermodynamic Favorability
Thermodynamic favorability is influenced by two main factors: enthalpy (ΔH) and entropy (ΔS). Generally, the enthalpy factor is more significant, but entropy becomes increasingly important at higher temperatures.
Enthalpy
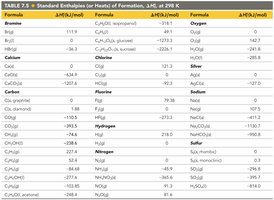
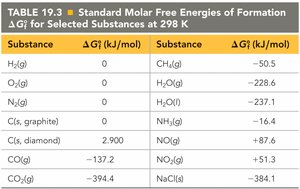
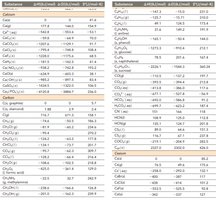
Standard Enthalpies of Formation
The standard enthalpy of formation (ΔHf°) is the enthalpy change when one mole of a compound is formed from its elements in their standard states. These values are used to calculate the enthalpy changes of reactions.
Entropy (S)
Definition and Properties
Entropy (S) is a measure of the dispersal of energy and the degree of disorder in a system. Systems with higher entropy have energy more widely spread among their particles.
Boltzmann Equation: where k is the Boltzmann constant ( J/K) and W is the number of microstates (ways to arrange the system).
Units: J/K
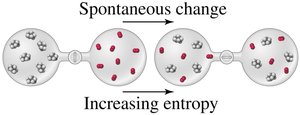
Natural Tendencies in Spontaneous Processes
There are two driving forces for spontaneous processes:
Tendency to achieve a lower energy state
Tendency toward a more disordered state (higher entropy)

Changes in Entropy (ΔS)
Entropy increases when a system becomes more disordered. Several types of changes increase entropy:
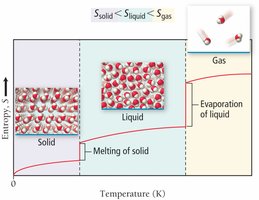
Phase changes: solid < liquid < gas
Dissolution of solids into ions
Reactions producing more product molecules than reactant molecules
Increase in temperature
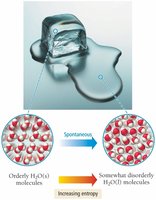
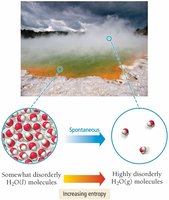
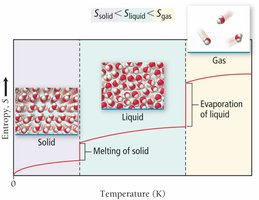

Entropy Change and State Change
Entropy always increases with temperature and increases dramatically during a phase change, such as melting or vaporization.
The Second Law of Thermodynamics
Statement and Mathematical Formulation
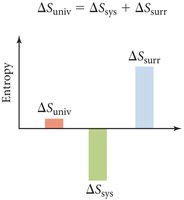
The Second Law of Thermodynamics states that the entropy of the universe increases for any spontaneous process:
For a spontaneous process:
Heat exchange with surroundings:
Gibbs Free Energy (ΔG)
Definition and Criteria for Spontaneity
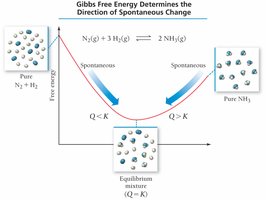
Gibbs free energy (ΔG) combines enthalpy and entropy to predict spontaneity at constant temperature and pressure:
If , the process is spontaneous.
If , the process is nonspontaneous.
If , the system is at equilibrium.
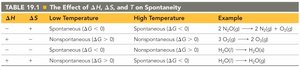
Effects of ΔH, ΔS, and T on Spontaneity
The sign and magnitude of ΔG depend on the values of ΔH, ΔS, and temperature (T). The table below summarizes the effect of these variables:
ΔH | ΔS | Low Temperature | High Temperature | Example |
|---|---|---|---|---|
- | + | Spontaneous (ΔG < 0) | Spontaneous (ΔG < 0) | 2 NH3 → N2 + 3 H2 |
+ | - | Nonspontaneous (ΔG > 0) | Nonspontaneous (ΔG > 0) | 2 H2O → 2 H2 + O2 |
- | - | Spontaneous (ΔG < 0) | Nonspontaneous (ΔG > 0) | H2O(l) → H2O(s) |
+ | + | Nonspontaneous (ΔG > 0) | Spontaneous (ΔG < 0) | H2O(s) → H2O(l) |
The Third Law of Thermodynamics and Absolute Entropy
Absolute Entropy
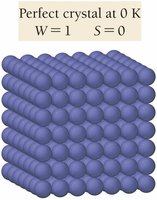
The Third Law of Thermodynamics states that the entropy of a perfect crystal at absolute zero (0 K) is zero. All substances have positive entropy values at temperatures above 0 K.
Standard Molar Entropies
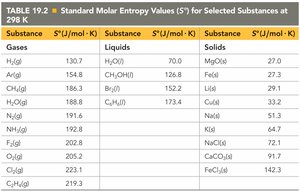
The standard molar entropy (S°) is the entropy of one mole of a substance in its standard state (1 atm, 25°C). Entropy values are always positive and increase with temperature.
Factors Affecting Entropy
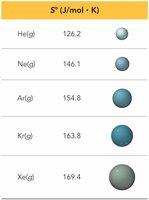
Physical State: S(solid) < S(liquid) < S(gas)
Molar Mass: Larger molar mass generally means higher entropy.
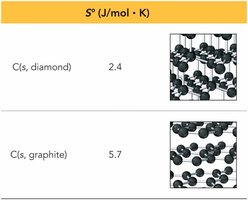
Allotropes: Less constrained structures have higher entropy.
Molecular Complexity: More complex molecules have higher entropy.
Dissolution: Dissolved solids have higher entropy than their undissolved forms.

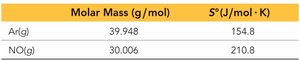
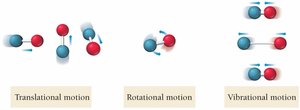

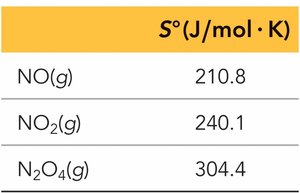

Calculating ΔS°, ΔH°, and ΔG° for Reactions
Standard State Calculations
Free Energy Change and Equilibrium
Relationship between ΔG, ΔG°, and Q
At equilibrium, and , so
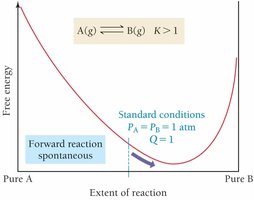
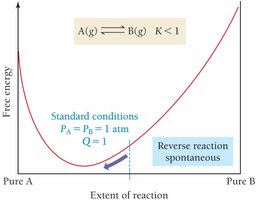
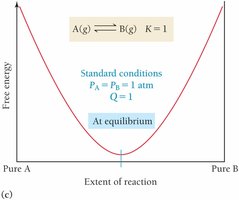
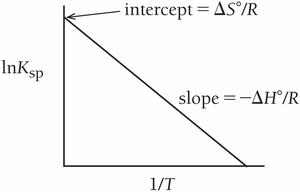
Temperature Dependence of Keq (van't Hoff Equation)
The van't Hoff equation relates the equilibrium constant to temperature:
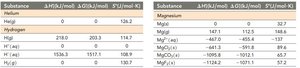
Standard Thermodynamic Quantities for Selected Elements
Summary Table: Key Equations
First Law:
Enthalpy:
Entropy (Boltzmann):
Second Law:
Gibbs Free Energy:
Relationship to Equilibrium:
At Equilibrium:
