 Back
BackFrom Amino Acids to Proteins: Structure, Bonding, and Properties
Study Guide - Smart Notes

From Amino Acids to Proteins
Introduction to Amino Acids
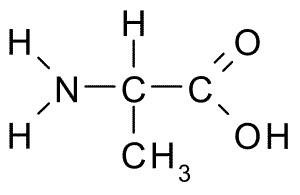
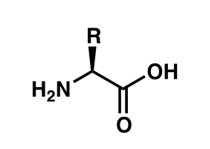
Amino acids are the fundamental building blocks of proteins, which are essential macromolecules in all living organisms. Each amino acid contains an amine group, a carboxyl group, a hydrogen atom, and a unique side chain (R group) attached to a central (alpha) carbon atom.
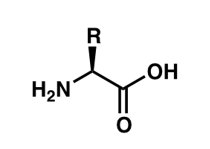
General Structure: The central carbon (Cα) is bonded to four different groups: an amino group (–NH2), a carboxyl group (–COOH), a hydrogen atom, and a variable R group.
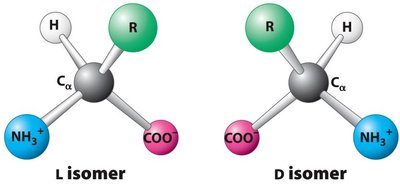
Chirality: With the exception of glycine, all amino acids are chiral and exist as L- and D-isomers. In biological systems, only L-amino acids are incorporated into proteins.
Functional Groups: The amine and carboxyl groups allow amino acids to link together via peptide bonds, forming polypeptides and proteins.
Classification of Amino Acids
Amino acids are classified based on the properties of their side chains (R groups):
Nonpolar (Hydrophobic): Side chains are mostly hydrocarbons, making them insoluble in water. Examples: Alanine, Valine, Leucine, Isoleucine, Methionine, Phenylalanine, Tryptophan, Proline, Glycine.
Polar (Hydrophilic): Side chains contain groups that can form hydrogen bonds with water, making them soluble. Examples: Serine, Threonine, Cysteine, Tyrosine, Asparagine, Glutamine.
Acidic: Side chains contain a carboxyl group, giving a negative charge at physiological pH. Examples: Aspartic acid, Glutamic acid.
Basic: Side chains contain an amino group, giving a positive charge at physiological pH. Examples: Lysine, Arginine, Histidine.
Peptide Bond Formation
Amino acids are linked by peptide bonds to form polypeptides and proteins. The peptide bond is a covalent bond formed between the carboxyl group of one amino acid and the amino group of another, releasing water (a condensation reaction).
Planarity: The peptide bond is planar due to resonance, restricting rotation and favoring either a trans or cis configuration (trans is preferred).
Directionality: Polypeptides have an N-terminus (amino end) and a C-terminus (carboxyl end).
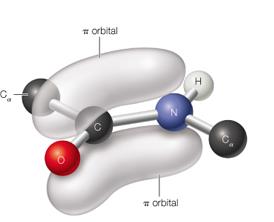
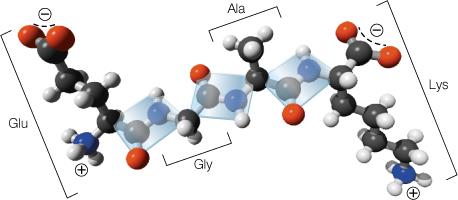
Bonding in Proteins: Sigma and Pi Bonds
Covalent bonds in proteins include sigma (σ) and pi (π) bonds:
Sigma (σ) Bonds: Electron density lies along the axis between two nuclei, forming single bonds.
Pi (π) Bonds: Electron density lies above and below the plane of the nuclei, present in double and triple bonds.
Resonance: The peptide bond exhibits resonance, giving partial double-bond character and restricting rotation.
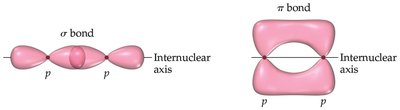
Levels of Protein Structure
Proteins have four levels of structural organization:
Primary Structure: The linear sequence of amino acids in a polypeptide chain.
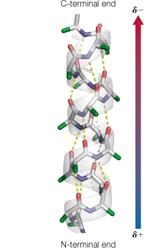
Secondary Structure: Local folding into α-helices and β-sheets stabilized by hydrogen bonds.
Tertiary Structure: The overall 3D shape of a single polypeptide chain, stabilized by various interactions (hydrophobic, hydrogen bonds, ionic bonds, disulfide bridges).
Quaternary Structure: The arrangement of multiple polypeptide subunits in a protein complex.
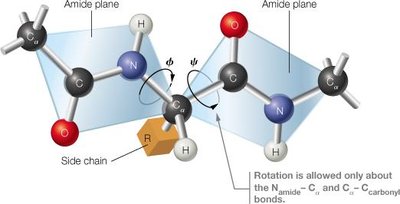
Protein Folding and Stability
Protein folding is driven by the sequence of amino acids and stabilized by several types of interactions:
Hydrophobic Effect: Nonpolar side chains aggregate to minimize contact with water, stabilizing the protein core.
Hydrogen Bonds: Form between backbone atoms and side chains, stabilizing secondary and tertiary structures.
Ionic Interactions (Salt Bridges): Occur between oppositely charged side chains.
Disulfide Bonds: Covalent bonds between cysteine residues, providing extra stability.
Van der Waals Interactions: Weak attractions between all atoms, significant when many are present.
Essential and Nonessential Amino Acids
Amino acids are categorized based on dietary requirements:
Essential Amino Acids | Nonessential Amino Acids |
|---|---|
Phenylalanine, Valine, Threonine, Tryptophan, Isoleucine, Methionine, Leucine, Lysine, Histidine | Arginine, Aspartic Acid, Glutamic Acid, Serine, Asparagine, Glutamine, Cysteine, Glycine, Proline, Alanine, Tyrosine |
Note: Essential amino acids must be obtained from the diet, while nonessential amino acids can be synthesized by the body.
Protein Misfolding and Disease
Improper protein folding can lead to aggregation and is associated with diseases such as Alzheimer's, Parkinson's, and Huntington's disease. Molecular chaperones assist in proper folding and refolding of proteins.
Chaperones: Proteins that help other proteins fold correctly and prevent aggregation.
Proteostasis Network: Cellular system maintaining protein homeostasis, involving chaperones, degradation pathways, and refolding mechanisms.
Summary Table: Types of Interactions in Proteins
Interaction Type | Description | Relative Strength |
|---|---|---|
Hydrophobic Effect | Aggregation of nonpolar groups to minimize water contact | Weak (cumulative effect is strong) |
Hydrogen Bond | Between electronegative atom and hydrogen bound to another electronegative atom | Moderate |
Ionic Bond (Salt Bridge) | Between oppositely charged side chains | Strong (in nonpolar environment) |
Disulfide Bond | Covalent bond between two cysteines | Very strong |
Van der Waals | Weak attractions between all atoms | Very weak (individually) |
Example: Alanine Structure
Alanine is a nonpolar, hydrophobic amino acid with the following structure:

Example: L- and D- Isomers
Amino acids can exist as L- or D- isomers, but only L-isomers are found in proteins:

Example: Peptide Bond Planarity
The peptide bond is planar due to resonance, restricting rotation and favoring the trans configuration:
Example: Protein Secondary Structure
Alpha helices and beta sheets are stabilized by hydrogen bonds:


Additional info:
Post-translational modifications such as phosphorylation can alter protein function and are important in cell signaling.
Protein folding is a complex process influenced by the sequence of amino acids and the cellular environment.
