 Back
BackGeneral Chemistry Exam 1 Study Guidance
Study Guide - Smart Notes

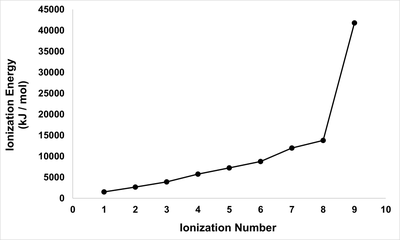
Q16. Consider the following plot of successive ionization energies for an element in the third period of the Periodic Table of Elements. What is the identity of this element?
Background
Topic: Periodic Trends – Ionization Energy
This question tests your understanding of how ionization energies change as electrons are removed from an atom, and how these trends can be used to identify elements based on their position in the periodic table.
Key Terms and Concepts:
Ionization Energy (IE): The energy required to remove an electron from a gaseous atom or ion.
Successive Ionization Energies: The energies required to remove each electron in sequence from an atom.
Electron Configuration: The arrangement of electrons in an atom, which determines how many electrons can be removed before a large jump in ionization energy occurs.
Core vs. Valence Electrons: Removing valence electrons requires less energy than removing core electrons, which causes a large jump in ionization energy after all valence electrons are removed.
Step-by-Step Guidance
Examine the plot and note where the largest jump in ionization energy occurs. This jump indicates that all valence electrons have been removed, and the next electron comes from a much more stable, inner shell (core electron).
Count the number of ionizations before the large jump. This number corresponds to the number of valence electrons in the atom.
Recall that elements in the third period are: Na, Mg, Al, Si, P, S, Cl, Ar. Match the number of valence electrons (from the plot) to the correct element in the third period.
Consider the electron configuration of each candidate element to confirm which one would show a large jump after removing its valence electrons.
Try solving on your own before revealing the answer!
